Principal Investigators:
Chris Leighton (IRG-1) and
Xiaojia Wang (IRG-1)
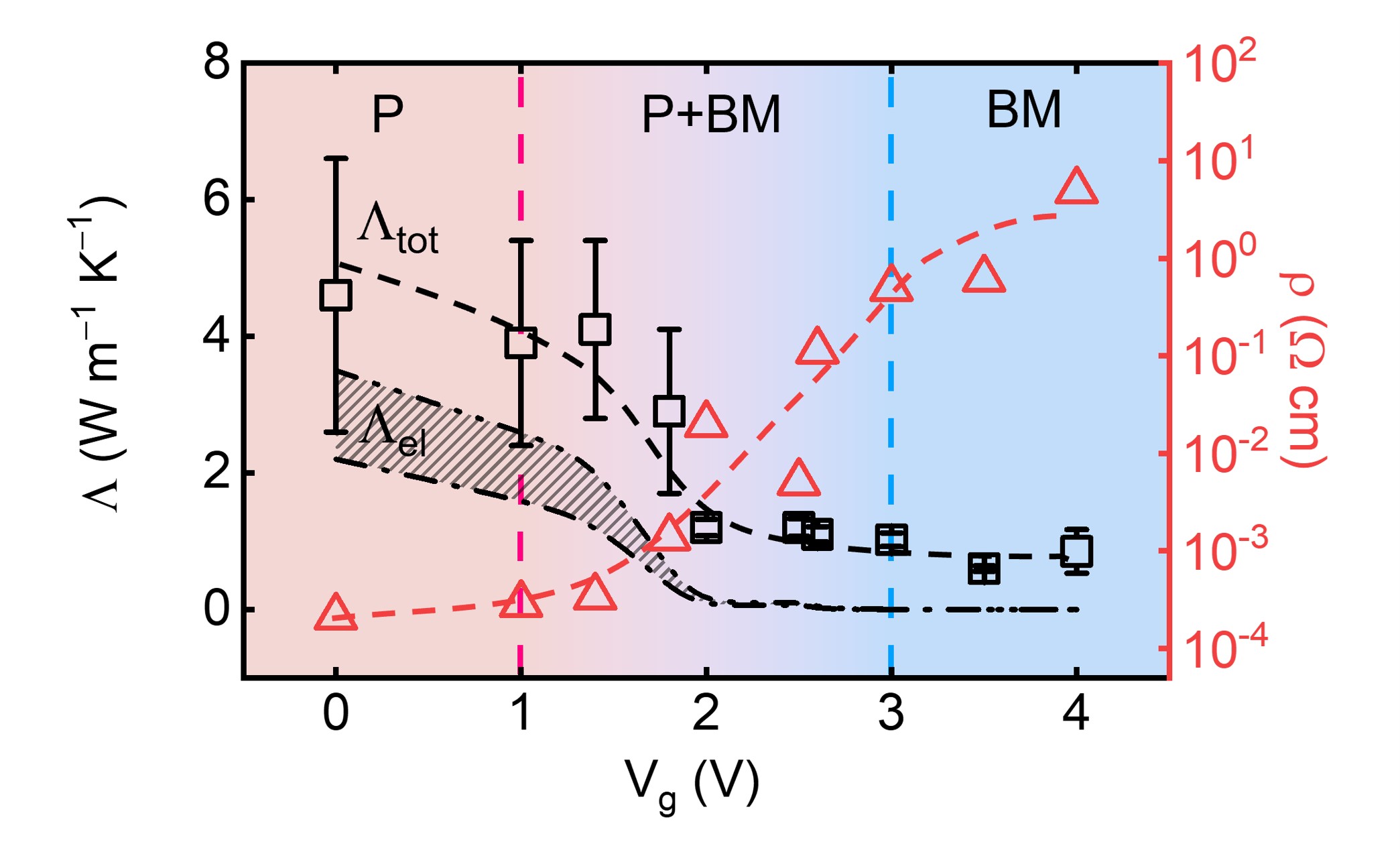
About the figure: Y. Zhang, W.M. Postiglione, R. Xie, C Zhang, H. Zhou, V. Chaturvedi, K. Heltemes, H. Zhou, T. Feng, C. Leighton, X.Wang, Nat. Commun., 14, 2626. (2023).
A team from IRG-1, working with collaborators at Argonne National Laboratory and the University of Utah, have demonstrated continuous room-temperature electrical tuning of the thermal conductivity of La0.5Sr0.5CoO3-δ by a factor of more than five (a record for a single-step process) via ion-gel gating. Application of a gate voltage in these devices drives a transformation from a metallic perovskite phase to an insulating brownmillerite phase via the formation and migration of oxygen vacancies, realizing the record range of measured thermal conductivities. The ability to control thermal conductivity electrically with a small voltage at room temperature is important for thermal regulation and management in several emergent device concepts.
The team’s approach used a combination of methods, including time-domain thermoreflectance, electronic transport, and X-ray diffraction at the Advanced Photon Source, Argonne National Laboratory, to measure and understand the mechanisms that enable this record tuning of the thermal conductivity.
Principal Investigators:
Frank Bates (IRG-2) and
Tim Lodge (IRG-2)
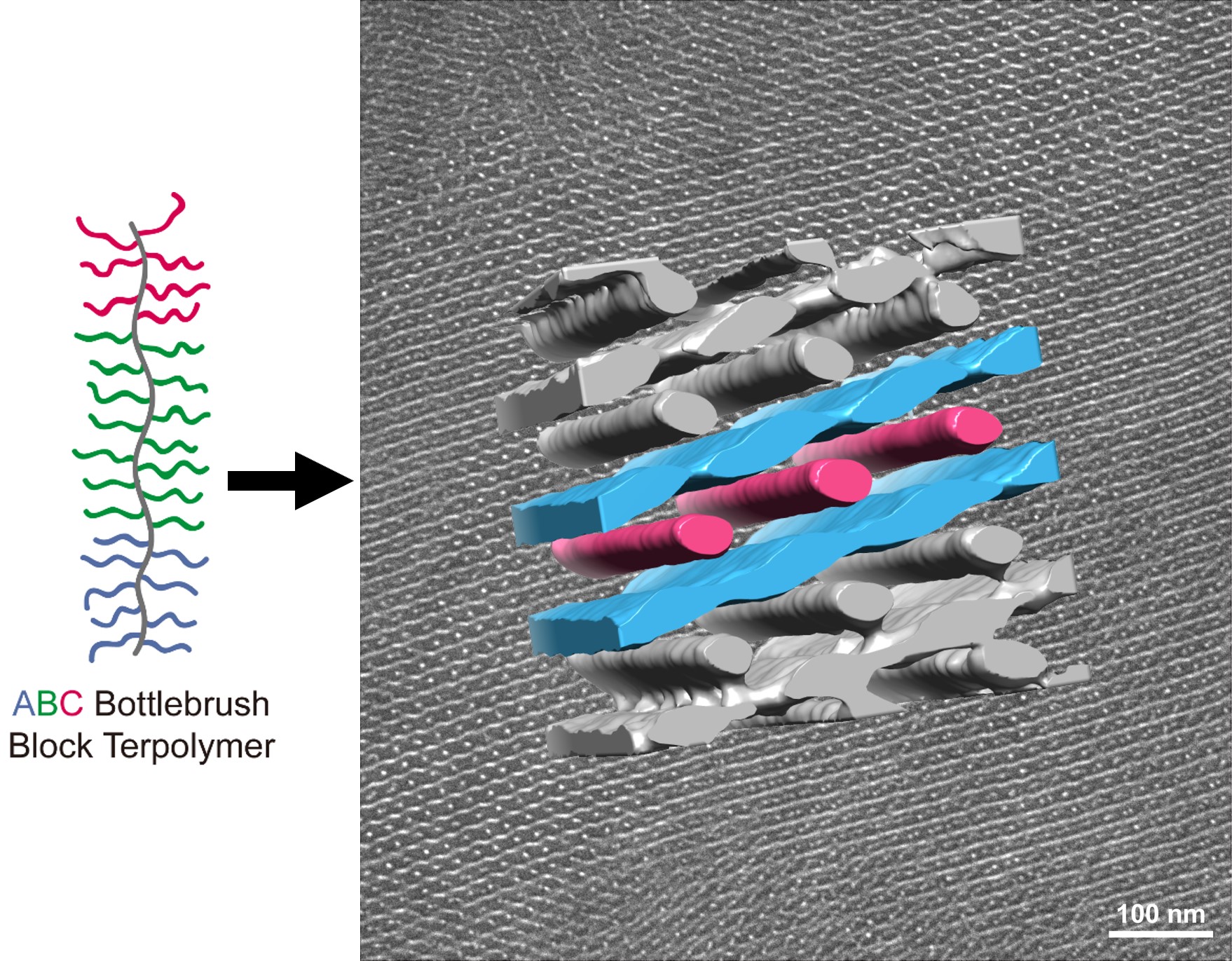
About the figure: S. Cui, E.A. Murphy, W. Zhang, A. Zografos, L. Shen, F.S. Bates, T.P. Lodge, J. Am. Chem. Soc., 146, 10, 6796-6805. (2024).
Soft materials known as molecular bottlebrush block polymers comprise a polymer backbone with densely grafted polymer side chains. These materials have attracted much attention for their ability to self-assemble into ordered structures with relatively large periodicities (over 50 nm), which are rarely achieved with simpler linear polymers. However, only self-assembly into lamellar and cylindrical phases has been reported in diblock bottlebrush materials. The absence of more complex self-assembled morphologies, which hold much potential for optical applications, may originate from the intrinsic stiffness of the bottlebrush polymer backbone.
To address this, IRG-2 researchers investigated the phase behavior of certain non-frustrated ABC bottlebrush block terpolymers. Surprisingly, an unprecedented cylinders-in-undulating-lamellae (CUL) morphology (see figure) was discovered, across a similar range of compositions where a network phase forms in linear triblock polymers. The effects of polymer dispersity on the CUL stability were scrutinized in collaboration with the Bio-Pacific MIP at UC Santa Barbara. This study demonstrates that stiff bottlebrush architectures with more than two blocks furnish exciting opportunities for the development of new materials with attractive nanoscale morphologies, and thus application potential in various areas.
Principal Investigators:
Andre Mkhoyan (IRG-1/Seed) and
Bharat Jalan (IRG-1)

About the figure: S. Guo, S. Nair, B. Jalan, A. Mkhoyan, Nat. Commun., 14, 6005. (2023).
Interactions between electrons and matter enable important atomic-resolution studies of materials with methods such as scanning transmission electron microscopy (STEM). These same interactions, however, also often result in sample degradation in the electron beam, via difficult to characterize mechanisms.
In this work, direct visualization of the restructuring of TiO2 under electron beam irradiation with annular-dark-field STEM imaging was achieved, revealing not degradation but instead progressive filling, i.e., repair, of atomically sharp nanometer-wide cracks, with atomic-resolution. Radiolysis was found to drive constructive changes in the atomic structure of TiO2, and STEM probes with varying beam energy and electron dose enabled quantification of the radiolytic mechanism. A “two-step rolling” model of mobile octahedral building blocks was discovered to underpin the radiolysis-driven atomic migration in this process. These results point to new possibilities for using electron beams to treat cracks in brittle ceramics, to improve the quality of wide bandgap thin film growth, and to engineer novel nanostructures with atomic precision.
.
Principal Investigators:
Turan Birol (IRG-1) and
Rafael Fernandes (IRG-1)
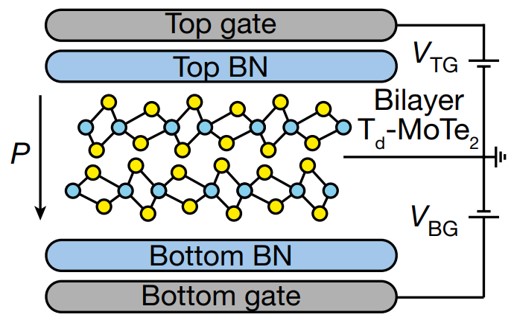
About the figure: A. Jindal, A. Saha, Z. Li, T. Taniguchi, K. Watanabe, J.C. Hone, T. Birol, R. M. Fernandes, C. R. Dean, A. N. Pasupathy, D. A. Rhodes, Nature, 613, 48-52. (2023)
For the first time, a team comprised of two IRG-1 theorists (Birol and Fernandes) working with experimentalists from other institutions (including the Columbia MRSEC) showed the coexistence of ferroelectricity (i.e., electrostatically switchable macroscopic dipole moment) and superconductivity in a two-dimensional superconductor. The material is bilayer MoTe2, a member of the family of transition metal dichalcogenides, which are compounds of high interest in IRG-1. This important result opens a new avenue to the control of superconductivity by electrostatic means.
The IRG-1 team’s theoretical approach combined first-principles and model calculations. Theoretical calculations were performed by IRG-1 student Amartyajyoti Saha. The theoretical analysis attributes the unique superconducting behavior of MoTe2 as a function of gating to an unconventional pairing state, thus shedding new light on the phenomenon of superconductivity in two-dimensional materials, of broad interest.
Principal Investigators:
Frank Bates (IRG-2) and
Kevin Dorfman (IRG-2)
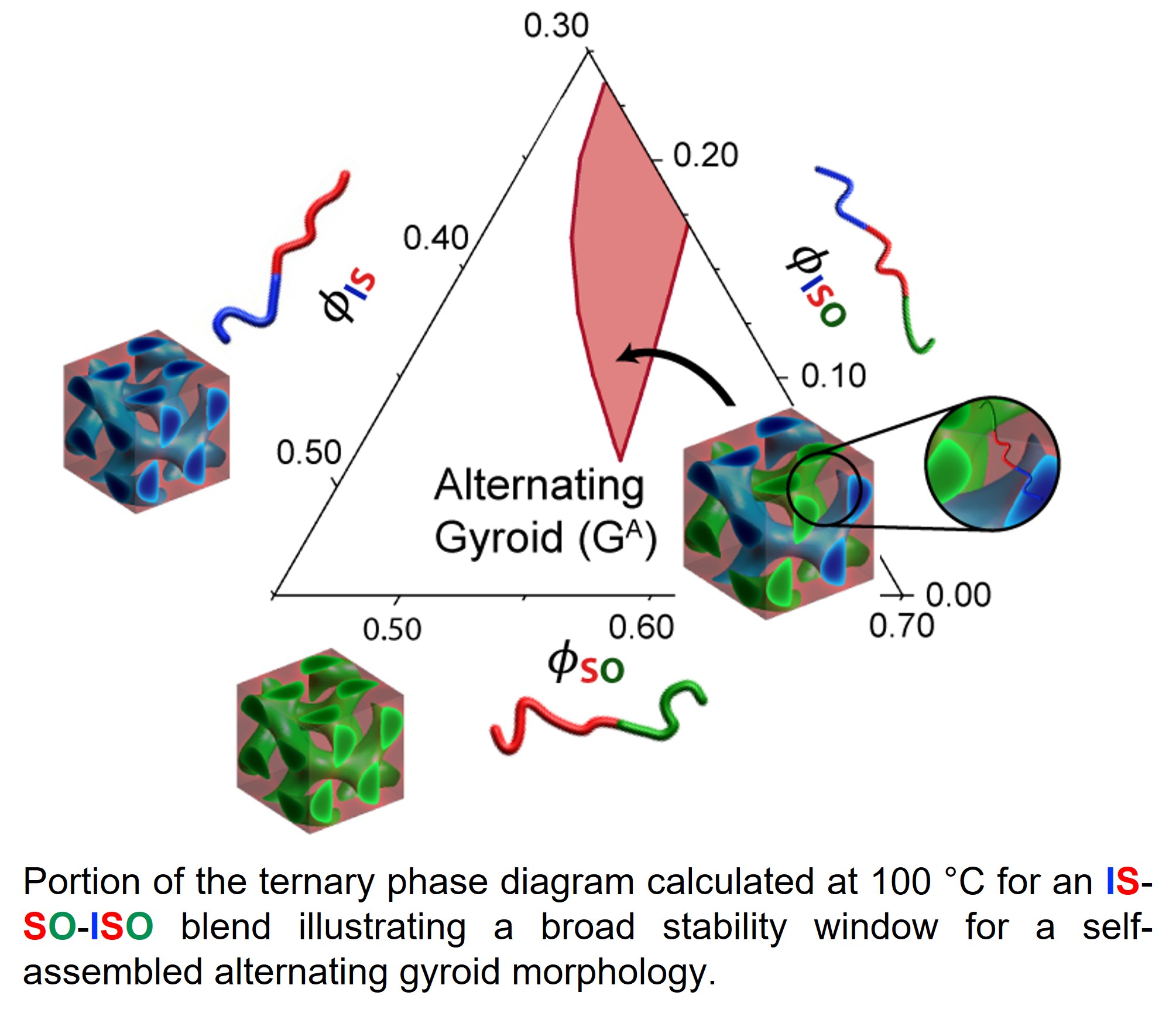
About the figure: P. Chen, F. S. Bates, K. D. Dorfman, Macromolecules, 56, 2568-2577. (2023)
An attractive approach to creating optical materials from self-assembled block copolymers is to create an alternating gyroid network (GA), then etch to remove one of the gyroids networks, followed by backfilling the void with metal to obtain a chiral material with high dielectric contrast.
IRG-2 researchers have used self-consistent mean-field theory to predict thermal and composition windows of alternating gyroid phase stability in experimentally-relevant blends of poly(isoprene) (I) , poly(styrene) (S) , and poly(ethylene oxide) (O) diblock and tri-block copolymers: IS, SO, and ISO (see figure). Owing to domain-size mismatches, the alternating gyroid stability window is shifted away from symmetric composition. Tuning both the blend composition and temperature minimizes the effect of the domain size mismatch. Since the I/S/O system is readily synthesized, the theoretical results set the stage for future experiments within the center to realize this material, and to study its processability and optical properties.
Principal Investigator(s):
C. L. Haynes (Seed)
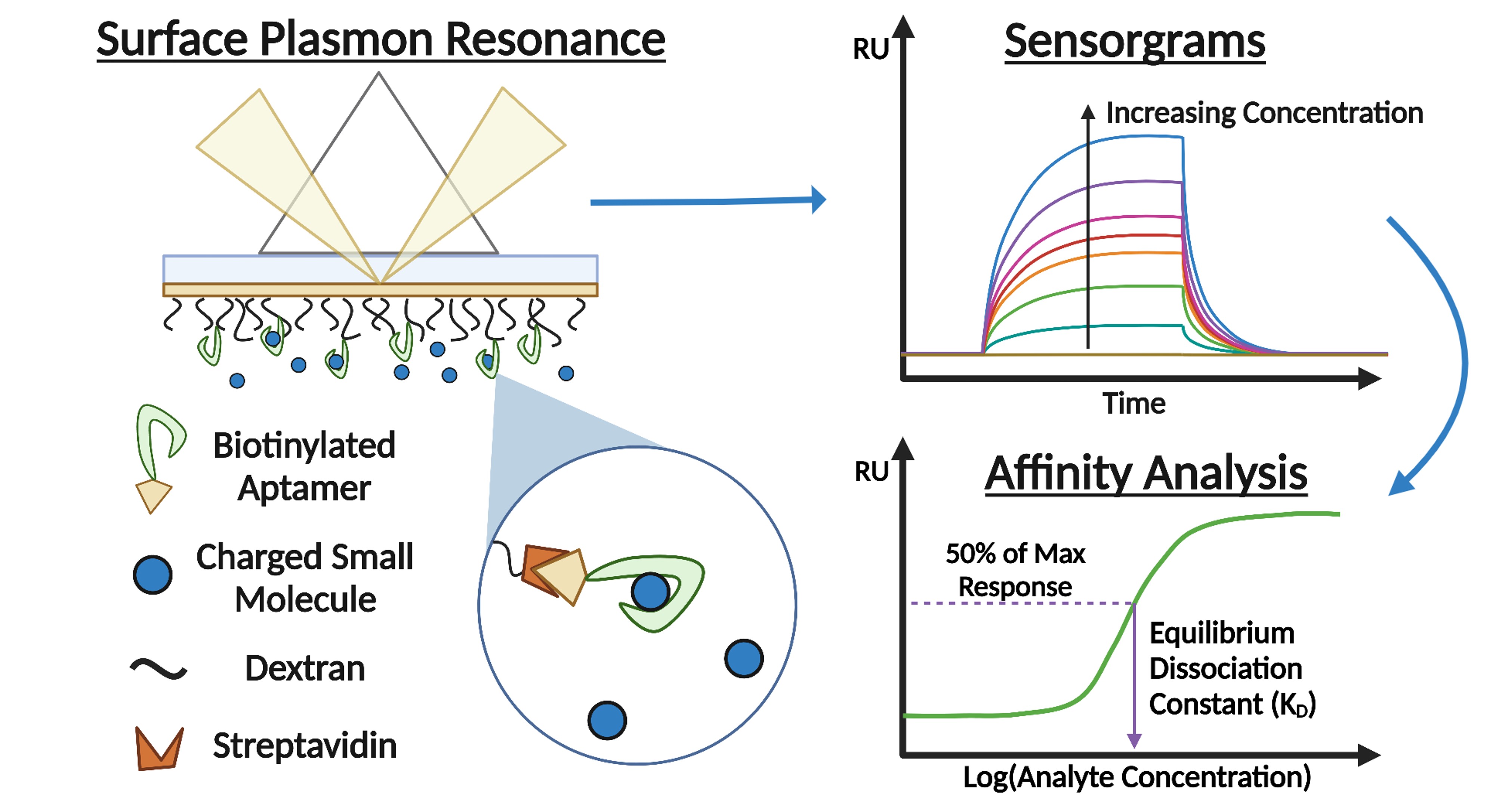
About the figure: C. E. Froehlich, J. He, C. L. Haynes, Anal. Chem., 95, 2639-2644. (2023)
A novel method for the study of interactions between small charged molecules and aptamers via surface plasmon resonance has been developed by MRSEC Seed experimentalist Haynes to inform collaborative work with Frisbie, Ferry (IRG-1), and Dorfman (IRG-2). The method focuses on controlling electrostatic interactions between the sensor surface, aptamer, and small molecule by using a high ionic strength buffer to screen charges and reduce repulsion.
The approach taken for method development, led by student Clarice Froehlich, involved exploration of multiple sensor surface functionalization techniques, all of which revealed information about how the small molecules were interacting with the material at the sensor surface.
As aptamers are commonly used to capture small molecules in electrochemical sensors, this development gives critical insight into how the sensor surfaces and materials can be designed for better sensing performance.
Principal Investigator(s):
M. Greven (IRG-1)

About the figure: M. Greven Lab, Quantum Design Inc., PPMS (2023)
In a critical shared instrumentation and infrastructure upgrade, a Quantum Design Inc. high-magnetic-field/low-temperature physical property measurement system (PPMS) was recently installed in the Greven Laboratory in the University of Minnesota (UMN) School of Physics and Astronomy. This versatile measurement platform is equipped with a 16-T superconducting magnet and a 50-mK-base-temperature dilution refrigerator insert. It will enable low-temperature/high-field electronic transport, heat capacity, torque magnetometry, and other measurements.
This 16-T/50-mK PPMS is the first system of its kind at a US university and provides the highest magnetic field and highest field/temperature ratio available at UMN. The system was acquired with a substantial MRSEC contribution in addition to internal UMN support. It was just installed and will be operated as an ISO (internal sales organization), making it accessible to all at UMN, including many MRSEC investigators, particularly in IRG-1 (Ionic Control of Materials). Ionically controlled materials exhibiting superconductivity and quantum oscillations are two specific targets for this new instrumentation.
Principal Investigators:
Chris Leighton (IRG-1) and
Turan Birol (IRG-1)
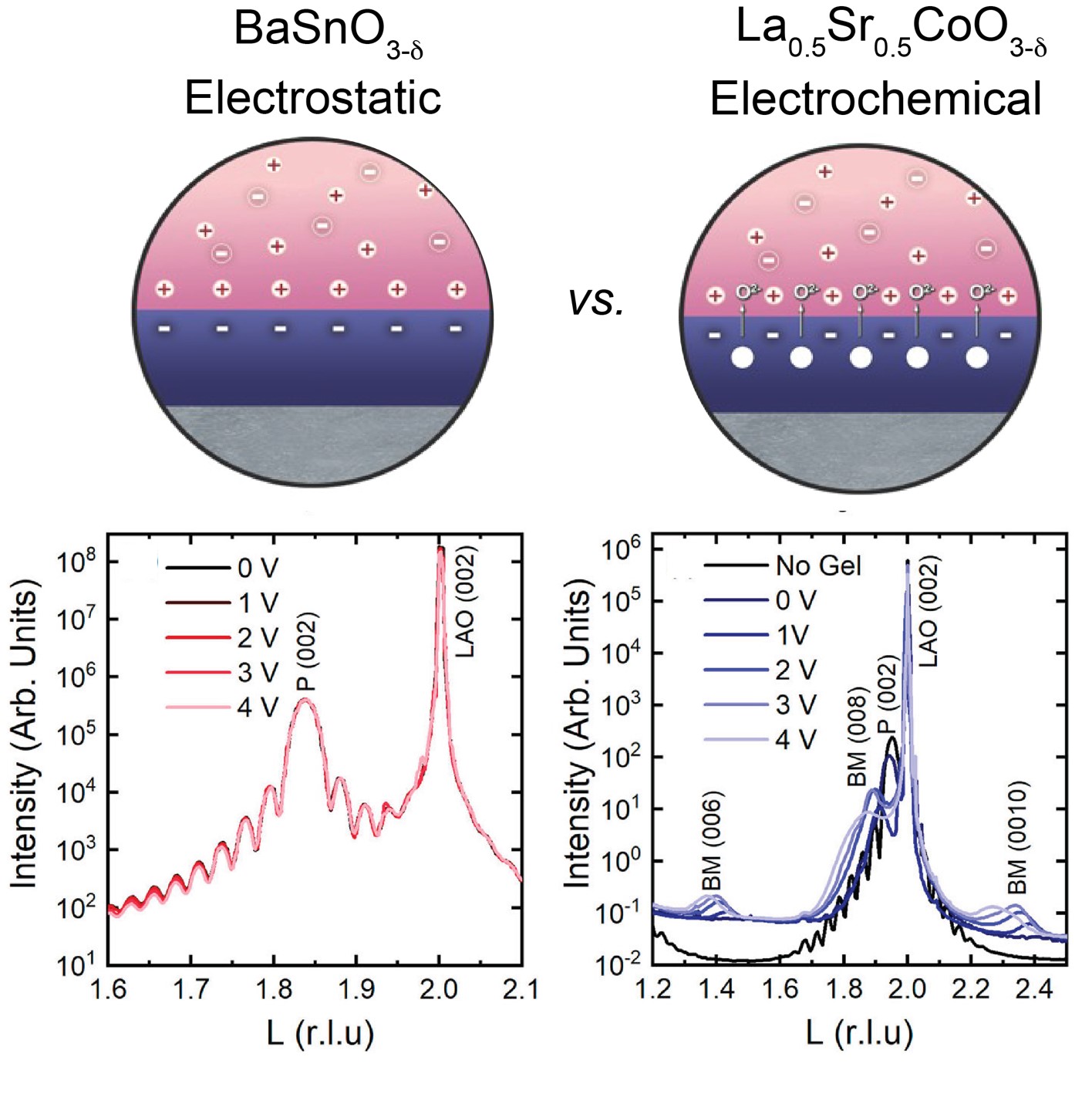
About the figure: C. Leighton, T. Birol, J. Walter, APL Mater., 10, 040901 (2022)
The electrolyte gating applied in IRG-1 is a powerful tool for control of a wide range of material properties, including superconductivity, magnetism, insulator-metal transitions, and optical properties. One of the puzzling observations, however, is that some closely related materials respond very differently to electrolyte gating. Specifically, some materials respond electrostatically, where electrons and holes are induced and ions move only through the electrolyte, while others respond electrochemically, where ions move on both sides (and across) the interface.
In this work, researchers in IRG-1 compared materials from the perovskite oxide and pyrite sulfide families that exhibit very different mechanisms upon gating to identify the precise materials factors that determine the mechanism. Surprisingly, it is proposed that it is the diffusivity of ions, rather than the energy to form them, that is critical to predicting the behavior under gating. This hypothesis can now be explored in a broader range of materials systems to realize control over numerous exciting materials properties.
Principal Investigators:
Frank Bates (IRG-2) and
Tim Lodge (IRG-2)
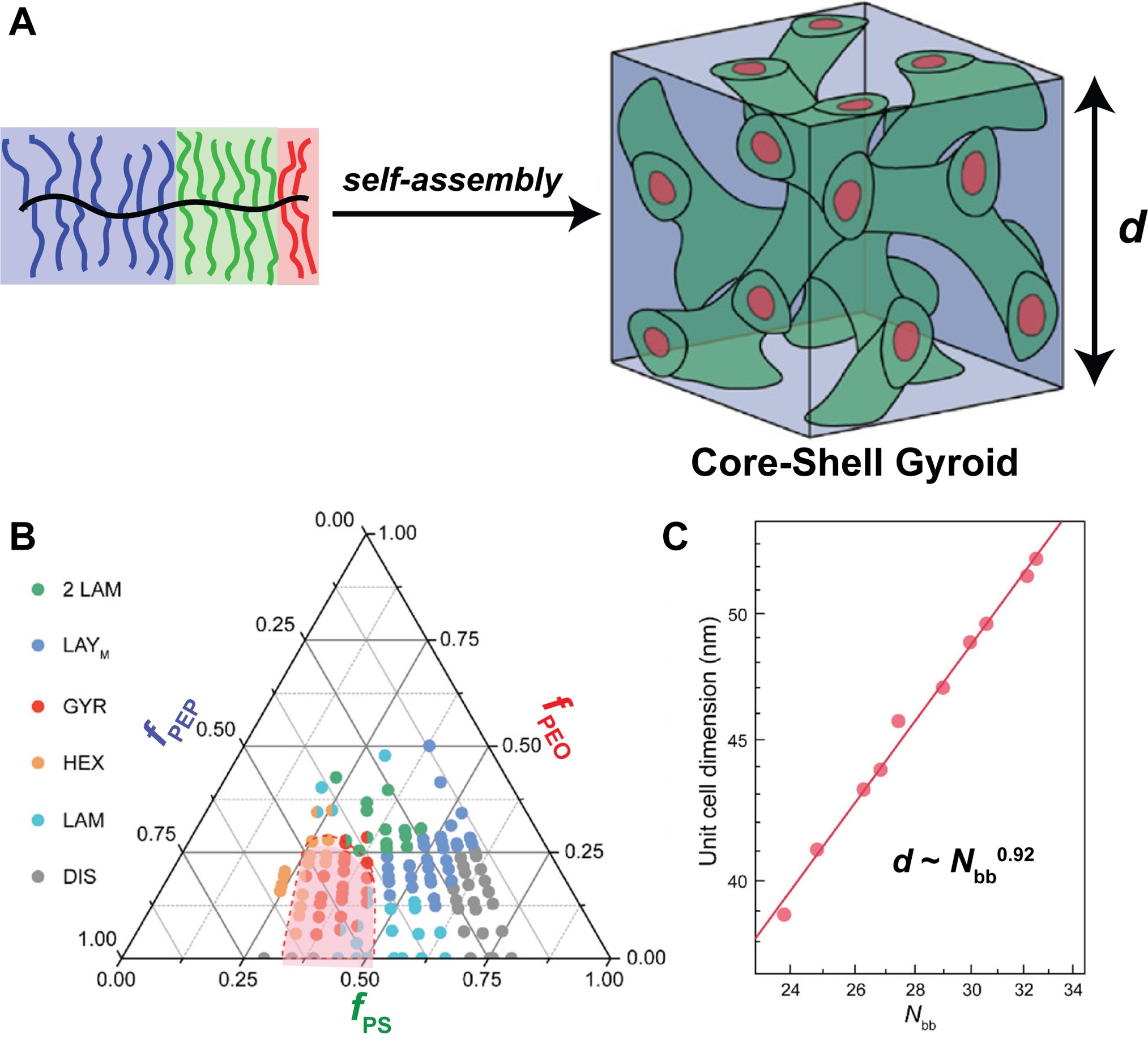
About the figure: S. Cui, B. Zhang, L. Y. Shen, F. S. Bates, T. P. Lodge, J. Am. Chem. Soc., 144, 21719-21727 (2022)
A principal obstacle to widespread applications of self-assembled network morphologies (NETs) of linear block polymers is access to only limited pore diameters and unit cell dimensions (typically <50 nm), originating from their coil configurations and slow self-assembly kinetics at high molar masses. A bottlebrush molecular architecture with extended backbone configurations due to side chain crowding could circumvent these shortcomings. However, NETs are very rarely observed in block bottlebrush polymers.
IRG-2 researchers systematically explored the phase behavior of 25 AB diblock and 108 ABC triblock bottlebrush polymers. A core-shell double gyroid NET was achieved in a substantial subset of the ABC triblock bottlebrush terpolymers for the first time. Encouragingly, the unit cell dimensions (d) of these assemblies increase almost linearly with molecular weight, portending the ability to access larger NET dimensions than previously obtained with linear block polymers for potential optical materials applications.
Principal Investigators:
Karen Lozano (IRG-2) and
Frank Bates (IRG-2) and
David Blank(E&O)

About the figure: J. F. Dewilde, E. P. Rangnekar, J. M. Ting, J. E. Franek, F. S. Bates, M. A. Hillmyer, and D. A. Blank, ACS Omega, 4, 2661-2668 (2019)
The University of Minnesota MRSEC has long supported the extraordinarily successful live outreach show Energy and U, which impacts tens of thousands of K-12 students annually. Through the PREM partnership with University of Texas Rio Grande Valley (UTRGV), this was recently developed as a Spanish language version, Energía Y TÚ, available both in live format and on film.
On November 12, 2022, the UMN and UTRGV teams behind the video adaptation of the Energy and U/Energía Y TÚ show were recognized with Lonestar Emmy awards in the Informational/Instructional short or long form content category. While many K-12 outreach activities, including the live version of Energy and U, were on hold due to COVID 19 restrictions, the team, led by Lozano (IRG-2), adapted the show to an interactive video format that students could participate in from their own homes or classrooms. The video version was presented in both English and Spanish to over 20,000 students in 2020-2021, providing critical STEM engagement during a time of relative isolation for K-12 students.

About the figure: University of Minnesota MRSEC, Education and Outreach
The University of Minnesota (UMN) RET Green and Sustainable Chemistry Workshop was devised as a way to extend the impact of the UMN MRSEC RET program. During the annual 3-day workshop in 2022, RET participants instructed 19 additional teachers on the lab- based classroom activities developed during the full 6-week UMN RET program.
The workshop had multiple sessions of both classroom and lab instruction. Participating teachers completed all lab experiments themselves to gain hands-on experience and were instructed on how each fits current curriculum and state standards in science and engineering. In the five years the workshop has run since 2017, nearly 100 teachers have participated. This has introducing RET-developed materials curriculum to thousands of students around the state of Minnesota, greatly leveraging the impact of the UMN RET program.
Principal Investigators:
Chris Ellison and
Vivian Ferry (IRG-2)
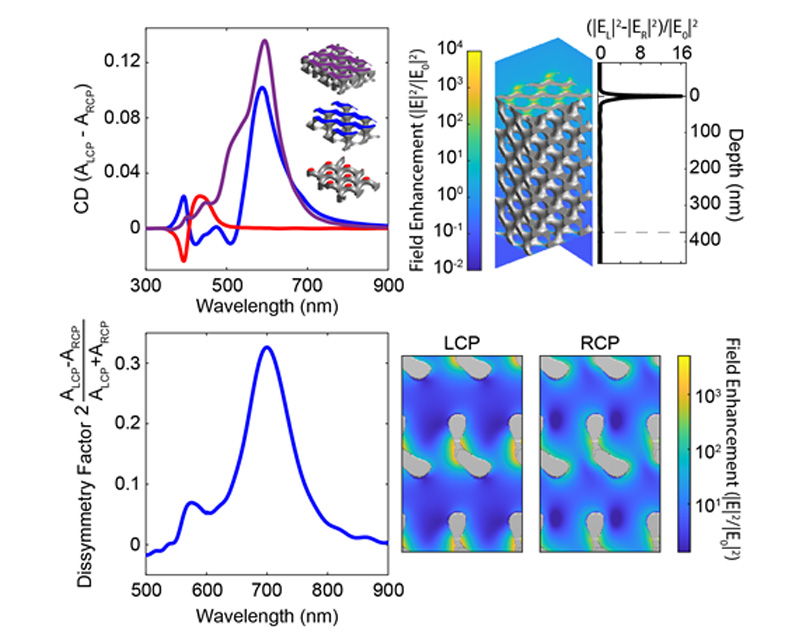
About the figure: B. M. Cote, W. R. Lenart, C. J. Ellison & V. E. Ferry, Adv. Optical Mater. 10, 2200363 (2022)
Materials with strong, selective interactions with circularly polarized light have wide-ranging applications, from biological detection to optical communications. Gyroid metamaterials are especially attractive candidates, since they may be fabricated by block copolymer self-assembly and templating. Using full-field electromagnetic simulations to study silver (Ag) gyroids of various orientations, we discovered the surface termination plane significantly influences the chiral optical response of the metamaterial. Judicious selection of the gyroid surface termination generates circular dichroism responses upwards of ±15% and Kuhn’s dissymmetry factors of nearly 0.33. Although the double gyroid is achiral in the bulk, the chirality of specific surface terminations supports strong chiral optical responses. Furthermore, surface roughness spreads the termination-dependent effects over a wider range of terminations, while the impact of defects in the bulk depends on their specific locations.
Principal Investigators:
Mahesh Mahanthappa,
Frank Bates,
Kevin Dorfman,
Tim Lodge,
and J. Ilja Siepmann (IRG-2)
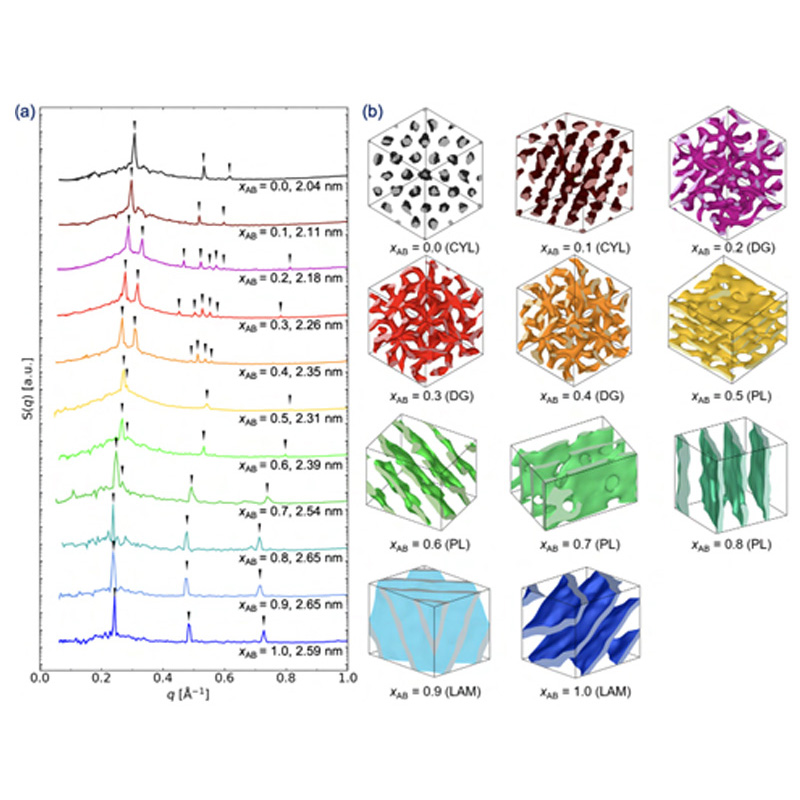
About the figure: Z. Shen, K. Luo, S. J. Park, D. Li, M. Mahanthappa, F. S. Bates, K. D. Dorfman, T. P. Lodge & J. I. Siepmann, JACS Au (2022)
Based on the hypothesis that blending LAM- and CYL-forming block oligomers may yield stable network phases, molecular dynamics simulations are
used to study binary blend self-assembly of AB-type diblock (n-tridecan-1,2,3,4-tetraol) and AB2-type miktoarm (5-octyl-tridecan-1,2,3,4-tetraol)
amphiphiles. The AB2-rich and AB-rich blends form double gyroid (DG) networks and perforated lamellae (PL), respectively. Structural
analyses reveal that the non-constant interfacial curvatures of DG and PL structures are supported by local composition variations of the LAM- and
CYL-forming amphiphiles.
Comparisons to self-consistent mean-field theory calculations reveal self-assembly differences and similarities between block copolymer blends and
stiffer, H-bonding oligomer mixtures, suggesting new design principles for tuning shape-filling oligomer and polymer architectures to achieve wide
stability windows for network phases.
Principal Investigators:
Frank Bates
and Tim Lodge (IRG-2)
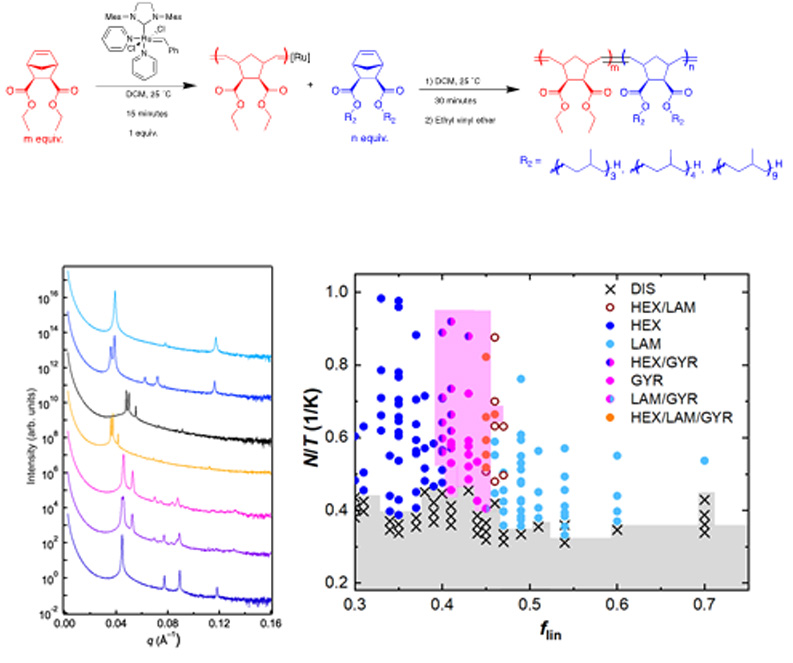
About the figure: L. Liberman, M. L Coughlin, S. Weigand, F.S. Bates and T. P. Lodge, Macromolecules, 55, 12 (2022)
We present a systematic study of architecturally asymmetric, linear-bottlebrush diblock polymers. Through synthesis of eight families, comprising 258
diblock polymers of varied compositions, molecular weights, architectural asymmetries, and linear block polarities, we assess double gyroid (DG) network
stability in linear-bottlebrush polymer melts.
By combining molecular and mesostructural characterization techniques, we reveal guiding principles that direct the self-assembly of these
architecturally asymmetric polymers into hexagonally-packed cylinders, DG, and lamellar mesophases. We show that the polymer architecture and chemical
identity of the linear block exert significant control over the compositions, compositional window width, and the molecular weight ranges over which
stable networks form.
Principal Investigators:
Mahesh Mahanthappa and
Kevin Dorfman
(IRG-2)

About the figure: P. Chen, M. K. Mahanthappa, K. D. Dorfman, J. Polym. Sci., 60, 2543-2552, (2022)
Double gyroid is the dominant network (Net) phase formed by diblock copolymer self-assembly, with a small O70 phase window near the order-disorder transition. However, many other Net phases are observed in nature, including iridescent insect wingscales.
Self-consistent field theory (SCFT) enables investigations of the relative free energies of these NETs, especially, the accessibility of metastable NETs through changes in materials processing protocols or molecular architecture. Conformational asymmetry furnishes a
simple approach to modeling molecular architecture changes, captured by the ratio ε between the stiffness of the majority block to the minority block. SCFT calculations of single and double gyroid (G), diamond (D), primitive (P), along with orthorhombic phases O52 and O70
and tricontinuous hexagonal H193 phases have revealed regions of the state space where novel NETs may be accessed by further advances in processing and molecular design.
Education & Outreach Highlight

The MRSEC Summer Undergraduate Research Expo (MRSEC SURE) is an exposition of undergraduate research for students from all over the country participating in summer research at the University of Minnesota and the surrounding area. In addition to the University of Minnesota campus which hosts hundreds of undergraduate researchers each summer, the Twin Cities is home to eight primarily undergraduate institutions (PUI) offering degrees in STEM fields- all within 10 miles of the UMN campus. MRSEC SURE offers those students the opportunity to gain experience presenting their research in a professional setting on a scale few would experience at their home institution. In addition to students, faculty, and researchers at all participating schools, students present their work to representatives from local companies invited to attend this public event. Despite reduced capacity due to the COVID pandemic and related restrictions, over 200 student posters were presented at the event on August 5, 2021.
Principal Investigators:
C. Daniel Frisbie (IRG-1) and international collaborators
About the figure: T. He, M. Stolte, Y. Wang, R. Renner, P. P. Ruden, F. Wurthner, C. D. Frisbie, Nat. Mater. 20, 1532-1538 (2021)
Doping - the deliberate introduction of impurities to control properties - is a mainstay of semi-conductor technology. While organic semi-conductors offer revolutionary possibilities such as cheap, low-energy-consumption fabrication, their doping is not well understood compared to conventional semiconductors. In this work, University of Minnesota MRSEC researchers and their international collaborators developed an entirely new site-specific chemical doping strategy for such semiconductors. This doping targets molecular steps on the surface of semiconducting crystals (see image), passivating them, chemically doping the material, and inducing massive improvements in electronic properties when electrostatically gated. The long-sought-after “band transport” was thus achieved even in challenging “n-type” molecular semiconductors, pointing to completely new research avenues, with much device potential.
Principal Investigators:
K. Andre Mkhoyan,
Bharat Jalan, and
Turan Birol (IRG-1)
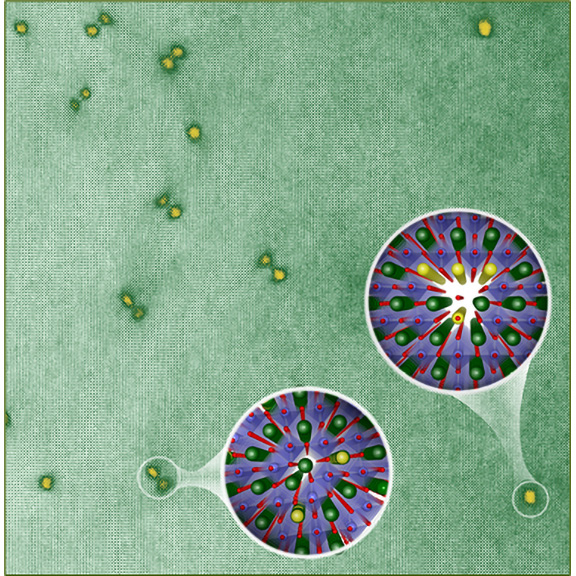
About the figure: H. Yun, A. Prakash, T. Birol, B. Jalan, and K. A. Mkhoyan, Nano Lett. 21, 4357 (2021)
Emerging semiconductors such as tin-based oxides have enormous application potential in devices, as they are transparent, support highly mobile electrons, and have wide “energy gaps”. Unlike better developed semiconductors, however, these materials are prone to harboring defects, which can limit essential properties such as electron mobility. In this work, University of Minnesota MRSEC researchers achieved the first detailed understanding of the interactions between the most important defects in these materials: deliberately-introduced “dopant” atoms, and naturally-forming “dislocations” (essentially extra chains of atoms). The dislocations were found to profoundly impact dopant atom locations, the formation of additional defects, and the electronic properties of the semiconductor, in unexpected ways. These results will now inform future studies to further refine the electronic properties of these exciting new materials.
Principal Investigators:
David Flannigan and
Ellad Tadmor (Seed)
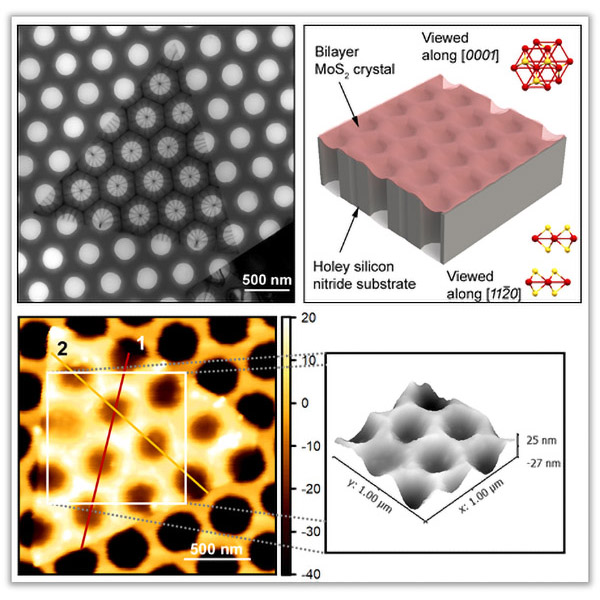
About the figure: Y. Zhang, M.-K. Choi, G. Haugstad, E. B. Tadmor and D. J. Flannigan, ACS Nano, (2021)
High strain tunability of 2D materials can be used to access new properties and functionalities (e.g., enhanced catalytic activities, tunable bandgap,
single-photon emission). Common approaches to apply strain include indirect manipulation of contact substrates and strain patterning using nanostructures,
but these methods require sustained loading or complex nanofabrication.
MoS2 is a prototypical 2D material with applications in wearable devices, photodetection, and next-generation transistors. We developed a method for
spontaneous strain patterning of bilayer MoS2 using commercially available holey substrates. The resulting highly symmetric strain patterns and surface
topography originate from out-of-plane, bowl-like 3D geometry of the crystal. Up to 35 nm of bending deformation and 1.8% strain were measured, and the
spontaneous formation of such topography was accurately predicted with atomistic simulations. This work enables a simple yet elegant method of fabricating
strain patterned 2D materials with bare, holey substrates.
Principal Investigators:
K. Andre Mkhoyan,
Turan Birol, and
Bharat Jalan
(IRG-1)
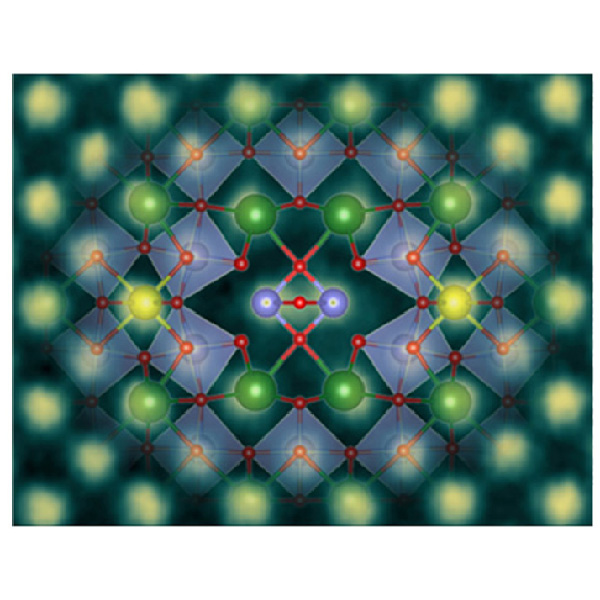
About the figure: H. Yun, M. Topsakal, A. Prakash, B. Jalan, J. S. Jeong, T. Birol, and K. A. Mkhoyan, Sci. Adv., 7, eabd4449, (2021)
For the first time a new kind of line defect with metallic characteristics has been discovered in optically transparent and insulating BaSnO3 perovskite thin films. It was identified using advanced atomic-resolution electron microscopy. The line defect has a very distinct atomic structure built up mainly from Sn and O atoms. When the electronic properties of the line defect were probed in the electron microscope, it was found to be metallic. Calculations based on theory from first principles confirm this unusual metallic property of the defect. This discovery demonstrates that extended line defects in crystals can accommodate remarkable properties and provide a unique way to engineer material properties at the sub-nanometer scale.
Principal Investigators:
Timothy Lodge and
J. Ilja Siepmann
(IRG-2)
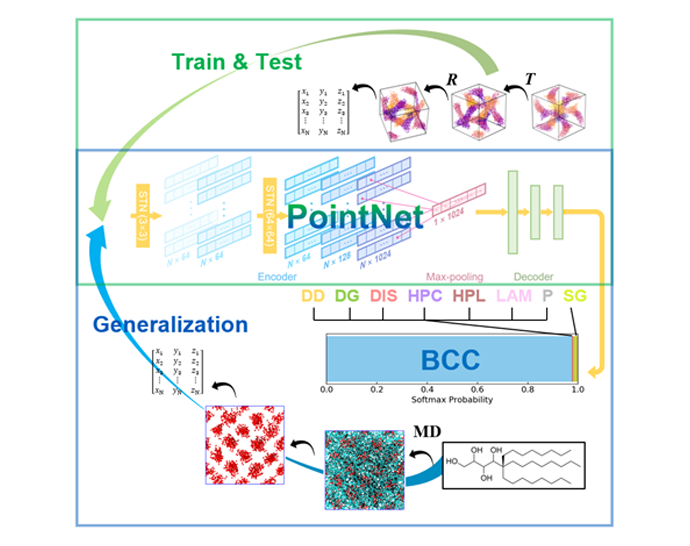
About the figure: Z. Shen, Y. Z. S. Sun, . P. Lodge & J. I. Siepmann, J. Phys. Chem. B, (2021)
Screening block oligomer chemistry and architecture through molecular simulations to find promising candidates for functional materials requires effective morphology identification techniques. Common strategies for structure identification include structure factors and order parameters, but these fail to identify imperfect structures in simulations with incorrect system sizes.
We developed a machine learning approach, called PointNet-meso, for mesophase classification including different network morphologies (double gyroid, single gyroid, double diamond, and plumber’s nightmare). The trained PointNet-meso model achieves an accuracy as high as 0.99 for globally ordered morphologies formed by linear diblock, linear triblock, and 3-arm and 4-arm star block oligomers, and it also allows for the discovery of emerging ordered patterns from non-equilibrium systems.
Principal Investigators:
Fiona Burnell,
Rafael Fernandes,
Ke Wang and
Vlad Pribiag
(iSuperSEED)
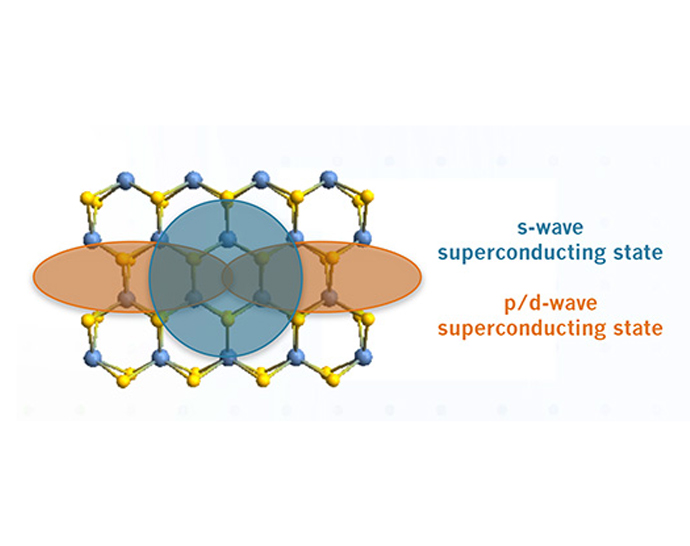
About the figure: A. Hamill, B. Heischmidt, E. Sohn, et al., Nature Physics, 17, 949-954, (2021).
Superconductivity is characterized by its ability to expel a magnetic field and conduct electricity with zero resistance. In particular circumstances, however, the way the superconducting state behaves goes against what is expected.
Here we report a superconducting state in NbSe2 that is stronger in one direction than another, despite expecting either no rotational variation (conventional s-wave state) or one that matches the three-fold rotational symmetry of the crystal.
This unexpected two-fold signal shows up as the material comes out of the superconducting state at both temperatures close to the transition (left panel) and close to absolute zero (right panel). After showing the signal’s nontrivial nature,
we propose that this effect arises from the interplay between two superconducting order parameters with different symmetries but that are close in energy. This study highlights the potential for exotic superconducting states in 2D van der Waals materials.
Education & Outreach Highlight

The University of Minnesota MRSEC’s commitment to Diversity and Inclusion is exemplified by the Summer Undergraduate Research Program. The program has worked to build long-term partnerships with faculty and administrators at Minority Serving Institutions (MSIs) as well as
student chapters of professional societies focused on women and minorities in STEM. The overall effect of this effort has been a steady increase in the participation of women and minority students over each previous award period (see panel a). Our initial cohort of
students in the current award (n=25) will have over 60% participation by both groups.
Our ultimate goal is to broaden participation in the STEM workforce. Over 95% of our highly diverse previous REU participants have entered careers in STEM and over 80% have enrolled in graduate programs. Graduate enrollment of MRSEC REU participants is also having a local impact.
In recent years, 70% of former REU participants enrolling in graduate programs at UMN, primarily in Materials Science or Chemical Engineering, have been women and 20% have been minority – both significantly higher than the overall matriculation rates in those
programs (32% and 5%, respectively; see panel b). With the strong start to the new award in 2021, the MRSEC REU program will continue to drive diversity in science and engineering both locally and nationally.
Principal Investigators:
Chris Leighton,
Turan Birol, and
Rafael Fernandes
(IRG-1)
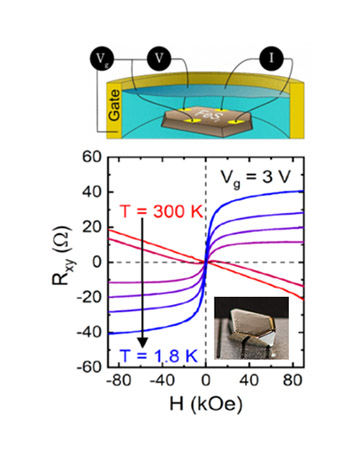
About the figure: J. Walter, B. Voigt, E. Day-Roberts, K. Heltemes, R.M. Fernandes, T. Birol and C. Leighton, Science Advances 6, eabb7721 (2020).
Dissipation of heat is a critical limiting factor in modern electronic devices such as transistors. Alternative technologies with the potential for low power consumption are thus of high interest, magnetoionics being one. In this emerging field, the goal is to direct the motion of ions with a voltage, but very little current, to control magnets. Recent research has yielded many advances, including voltage-induced ferromagnetism in various types of magnetic materials, but never in an entirely nonmagnetic material. In this work, exactly this feat was achieved by demonstrating that an approach called ionic liquid gating can voltage induce ferromagnetism in diamagnetic (i.e., nonmagnetic) FeS2 (“Fool’s Gold”). This result has very broad implications, potentially increasing the available palette of useful magnetic materials and enabling entirely new device function of use in technology.
Principal Investigators:
Mahesh Mahanthappa,
Theresa Reineke,
Tim Lodge,
and J. Ilja Siepmann (IRG-3)
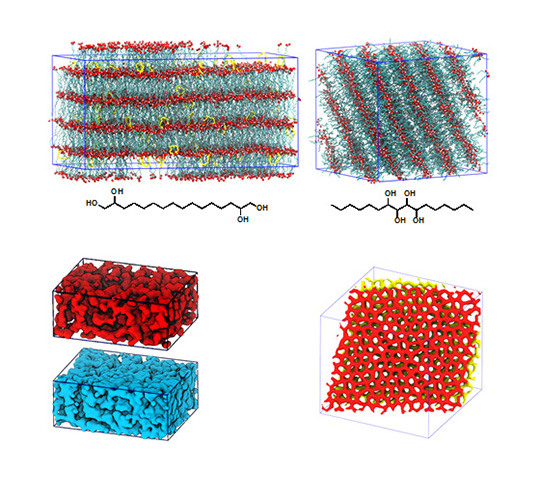
About the figure: from Z. Shen, J. L. Chen, V. Vernadskaia, S. P. Ertem, M. K. Mahanthappa, M. A. Hillmyer, T. M. Reineke, T. P. Lodge and J. I. Siepmann, J. Am. Chem. Soc., DOI: 10.1021/jacs.0c01829, (2020)
Block polymers are a class of versatile self-assembling soft materials that can form exquisite nanostructures for applications including ion transport membranes for batteries and fuel cells, and templates for inorganic oxide catalysts. Using molecular dynamics simulations and transferable force fields, we designed a series of symmetric triblock amphiphiles (or high-χ “block oligomers”) comprising incompatible sugar-based (A) and hydrocarbon (B) blocks that can self-assemble into ordered nanostructures with full domain pitches as small as 1.2 nm. Depending on the chain length and block sequence, the ordered morphologies include lamellae (LAM), perforated lamellae, and hexagonally-perforated lamellae (HPL). Above the order-disorder transition temperature, the disordered states are locally well-segregated and form bicontinuous network phases. This study furnishes detailed insights into structure-property relationships for mesophase formation on the 1-nm length scale that will aid further miniaturization for numerous applications.
Principal Investigators:
Theresa Reineke, Frank Bates,
and Tim Lodge (IRG-3)
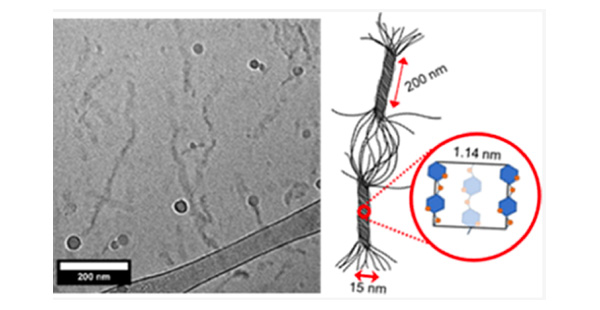
About the figure: from P. W. Schmidt, S. Morozova, S. P. Ertem, M. L. Coughlin, I. Davidovich, Y. Talmon, T. M. Reineke, F. S. Bates, T. P. Lodge, Macromolecules, 53, 398-405 (2020)
Methylcellulose (MC) is a water-soluble cellulose derivative with a wide range of commercial applications. Upon heating, MC solutions reversibly form ∼15–20 nm diameter fibrils, which percolate into a fibrillar network, resulting in macroscopic gelation. Using mid-angle X-ray scattering (MAXS) and wide-angle X-ray scattering (WAXS), we have analyzed MC chain organization within fibrils aligned in dried films that have been stretched by over 300%. MAXS and WAXS show distinct anisotropic scattering features, which we interpret as reflecting crystalline domains within the fibrils. Phase-plate cryogenic transmission electron microscopy images of MC fibrils contribute to a more comprehensive picture. Along the long axis of MC fibrils, there is evidence of dense twisted domains, which are interpreted as regions containing semicrystalline MC, interspersed with looser, less organized amorphous domains. Together, these two techniques provide the most complete interpretation of MC subfibril structure currently available.

About the figure: from J.F. DeWilde, E.P. Rangnekar, J.M. Ting, J.E. Franek, F.S. Bates, M.A. Hillmyer, and D.S. Blank, ACS Omega, 2019, 4 (2), 2661-2668. DOI: 10.1021/acsomega.8b03156.
Energy and U is an interactive demonstration show developed by MRSEC faculty at the University of Minnesota. Energy and U uses large-scale demonstrations, music,
and dance to teach the first law of thermodynamics and to illustrate global energy challenges. With an annual attendance of over 12,000 students, the show has now
been enjoyed by well over 100,000 students, primarily in grades 3-6.
The unique focus of the Energy and U show on teaching the 1st law of thermodynamics offered an opportunity to evaluate its immediate and long-term impact on student learning
and interest in STEM. Key findings from the evaluations, which were recently published in ACS Omega,1 showed that students were not only learning the key concepts presented
during the show, but retaining that information over time. Interestingly, the same results were shown for their adult chaperones. In addition, the assessment showed that
for this elementary age audience, socioeconomic factors were not significant in terms of future interest in attending college or general interest in STEM.
Principal Investigators:
Russell J. Holmes
and Laura Gagliardi (Seed)
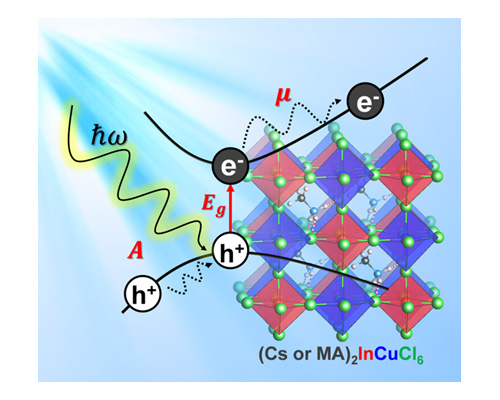
About the figure: from H. Q. Pham, R. J. Holmes, E. S. Aydil and L. Gagliardi, Nanoscale 11, 11173 (2019).
Lead (Pb) halide perovskites with the chemical formula APbX3, where A is a cation (e.g., cesium Cs+ or methyl ammonium CH3NH3+) and X is a halide (e.g., Cl–, Br–, or I–), are being vigorously explored as materials for low-cost and high efficiency solar cells (up to 23.3%, as reported in 2019). Pb-containing perovskites, however, face two problems: their limited chemical stability, and the negative impact of lead on the environment and human health. In this work, University of Minnesota investigators explored the properties of indium-based inorganic double perovskites Cs2InMX6 with M = copper, silver, or gold, using density functional theory. As a result, two new perovskites, Cs2InCuCl6 and (CH3NH3)2InCuCl6, are predicted to have promising characteristics, such as remarkable optical properties and high carrier mobilities. This work not only proposes two lead-free perovskites for light harvesting, but also highlights the importance of theoretical modeling in designing novel materials.
Principal Investigators:
Steve Koester,
Tim Lodge and
Dan Frisbie (IRG-1)
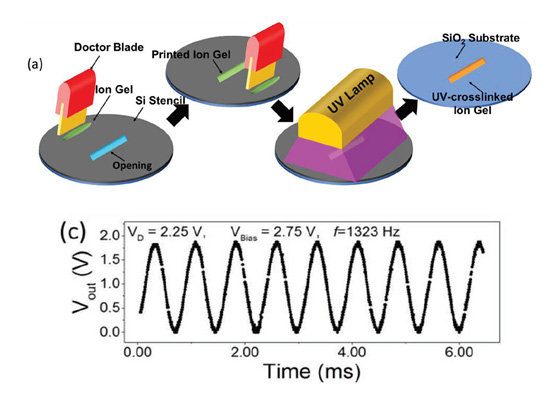
About the figure: from F.Z. Bidoky, B. Tang, R. Ma, K.S. Jochem, W.J. Hyun, D. Song, S.J. Koester, T.P. Lodge, and C.D. Frisbie, Adv. Func. Mater. 1902028 (2019).
An active frontier in science and engineering is the extension of the realm of microelectronic devices (e.g., transistors) to printed electronics. Success in this endeavor would enable low-cost high-throughput manufacturing of flexible, stretchable, even wearable electronics, with a multitude of applications. Investigators in MRSEC IRG-1 at the University of Minnesota are studying a particularly promising approach to printed transistors known as “electrolyte gating”. While powerful, this approach is typically employed at large size scales and modest frequencies, which are incompatible with printed electronics. In this work, an IRG-1 team synthesized a novel ion gel electrolyte to realize high-performance electrolyte-gate transistors down to micron sizes, operating at 20 kHz frequencies and low voltage. The work points to a clear route to even MHz frequencies, opening up a plethora of new applications.
Principal Investigators:
Chris Leighton and
Dan Frisbie (IRG-1)
About the figure: from X. Ren, Y. Wang, Z. Xie, F. Xue, C. Leighton and C.D. Frisbie, Nano Lett. 19, 4738 (2019)
Atomically-thin sheets of semiconductors have been of immense interest since the Nobel-Prize-winning discovery of graphene or two-dimensional (2D) carbon. Such materials represent the ultimate limit of “scaling” to small sizes, of vital importance in the semiconductor device industry. A particularly exciting recent (2017) finding is that the elemental semiconductor tellurium can be created in 2D sheets, with highly mobile electrons. In this work, University of Minnesota investigators in MRSEC IRG-1 applied “electrolyte gating” to this new material, using the motion of ions to control electronic properties. A transition from semiconductor to metal was found, enabling mapping of the electronic properties of “tellurene” over an extraordinary range. This work highlights not only the potential of tellurene for electronic devices, but also the general power of electrolyte gating (an IRG-1 focus) in 2D materials research.
Principal Investigators:
Uwe Kortshagen and
K. Andre Mkhoyan (IRG-2)
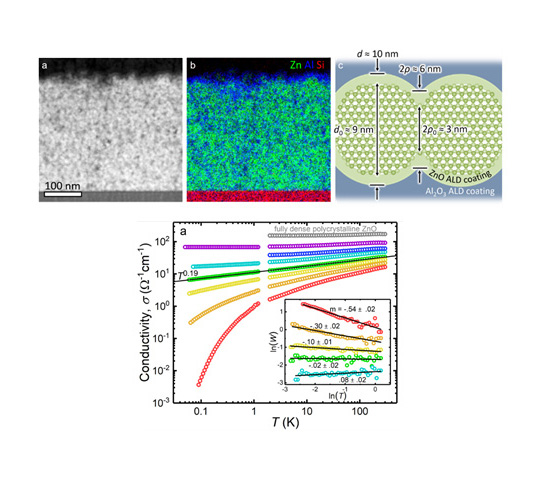
About the figure: An insulator-to-metal transition was demonstrated in a network of zinc oxide nanocrystals. (2019) DOI: 10.1126/sciadv.aaw1462
While nanocrystal films can be rapidly deposited from the gas phase on flexible substrates, achieving good electrical conductivity in these films has long remained a challenge. A multi-disciplinary IRG-2 research team demonstrated for the first time that the electrical transport in plasma-produced zinc oxide nanocrystal films can be enhanced to such a degree that the films start to behave like thin metal films. The team used a combination of gas-phase nanocrystal deposition, atomic layer deposition to seal the nanocrystal surfaces, and photodoping to achieve persistently enhanced electrical conductivities. Metallic behavior was indicated by conductivities that remained larger than zero even at temperatures as low as 50 milli-Kelvin. This study was reported in Science Advances.
Principal Investigators:
Xiaojia Wang (IRG-2)
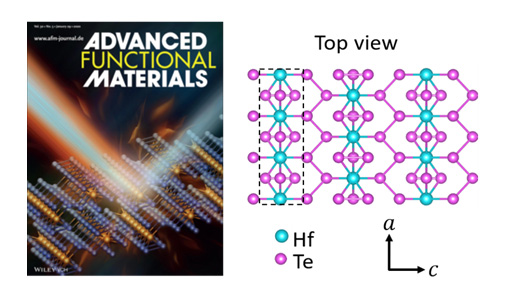
About the figure: from T.L. Feng, X.W. Wu, X.L. Yang, P.P. Wang, L.Y. Zhang, X. Du, X.J. Wang, and S.T. Pantelides, Adv. Funct. Mater. 30, 1907286 (2019).
Transition-metal pentatellurides have attracted extensive interest due to their exotic electronic, optical, and thermal properties. In this work, researchers in IRG-2 explored thermal transport in hafnium pentatelluride (HfTe5) and zirconium pentatelluride (ZrTe5). Both compounds are asymmetrical crystals (layered structures with in-plane quasi-1D chains), leading to the highly anisotropic thermal transport with an ultralow thermal conductivity along the interlayer direction (b-axis). Compared with ZrTe5, HfTe5 has heavier molar mass and thus is expected to have even lower thermal conductivity. However, combining high-fidelity ultrafast pump-probe thermal measurements and accurate theoretical calculations, researchers were able to show that the thermal conductivity of HfTe5 along the b-axis is higher than that of ZrTe5, due to its weaker anharmonicity. This work can provide guidance for the design of new directional-heat-management and high-efficiency thermal insulators.
Principal Investigators:
K. Andre Mkhoyan (IRG-2)
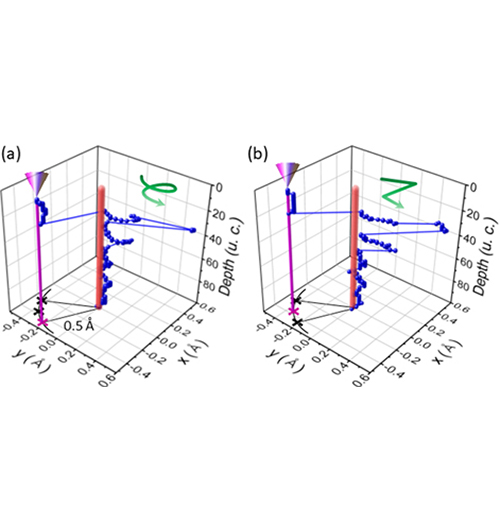
About the figure: from J. S. Jeong, H. Song, J. T. Held, K. A. Mkhoyan, Phys. Rev. Lett. 122 (2019). DOI: 075501.10.1103/PhysRevLett.122.075501
Using analytical aberration-corrected scanning transmission electron microscopy (STEM), we studied the behavior of the electron probe propagating in SrTiO3 at sub-atomic length scales. For the very first time, we discovered the unusual nature of sub-atomic beam channeling in crystalline material. In some cases electrons of the probe channel along the atomic column with pendulum-like motion, moving side-to-side inside the atoms. In the other cases, they spiral within the atoms forming a helicon-type beam. In the figure, the position of the peak intensity of the propagating beam (in unit cells, u.c.) located near the oxygen atomic column is shown for these two cases. This discovery suggests that there is a rich sub-atomic science to be explored using these beams. Atomic-scale magnetism is one field that can benefit dramatically from this discovery of spiraling electron beams in crystals.
Principal Investigators:
Tim Lodge (IRG-3)
Theresa Reineke (IRG-3)
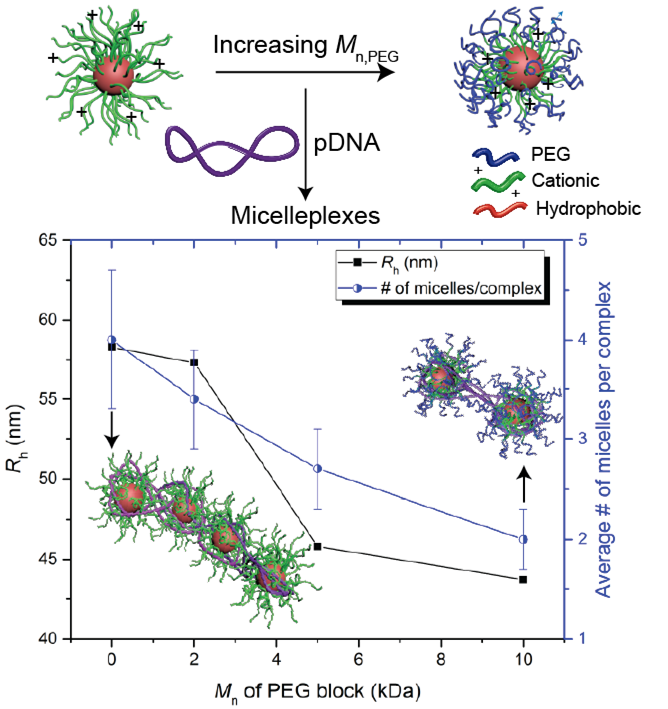
About the figure: (Top) Cationic micelles can be formed via self-assembly from amphiphilic block polymers and complexed with pDNA to form micelleplexes. (Bottom) The number of micelles per micelleplex was estimated using the Mw of the micelleplexes measured by SLS with the assumptions that all micelleplexes are identical and that there are no excess free micelles. Y. Jiang, T. P. Lodge, T. M. Reineke, J. Am. Chem. Soc. 140, 11101-11111 (2018). DOI: 10.1021/jacs.8b06309
The versatility of polyelectrolyte complexes resides in the facile incorporation of multiple chemical components in the composing polyions, which allows design of complexes with intricate structures and multiple functionalities. In this work, the complexation of ABC micelles with a model semiflexible polyion, DNA, is systematically investigated to correlate the structure of the micelle with the properties of the resulting “micelleplexes”. With the addition of a nonionic outer corona, the colloidal stability of the resulting micelleplexes dramatically improves and is maintained over a wide range of charge ratios. At the same time, the size, composition and structure of micelleplexes can be tuned by varying the nonionic outer corona length without compromising the colloidal stability. These results demonstrate a new strategy to compact long chain polyions and design nanoparticles with superior colloidal stability and tunable composition.
Principal Investigators:
Chris Leighton (IRG-1)
Rafael Fernandes (IRG-1)
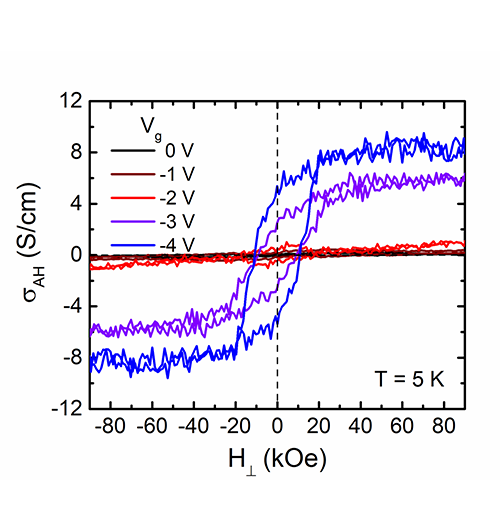
About the figure: from Jeff Walter, T. Charlton, H. Ambaye, M.R. Fitzsimmons, Peter P. Orth, R.M. Fernandes, and Chris Leighton, Phys. Rev. Materials. 2, 111406 (R) (2018). DOI: 10.1103/PhysRevMaterials.2.111406
Electrical control of magnetism is long sought-after in spintronics, potentially enabling new approaches to energy-efficient memory and logic chips. In IRG-1, researchers are addressing this challenge by applying an approach termed “electrolyte gating” to control magnetic properties by application of a voltage. The most recent development is successful implementation of a theoretical idea to enable large tunability in Curie temperature, i.e., the temperature at which magnetism turns on. By electrically connecting magnetic clusters in compounds known as perovskite oxides, an unusually large change in Curie temperature was obtained, up to 150 degrees with 4 volts, setting new records. The effect does not involve motion of ions in the oxide, potentially maximizing switching speed, important for applications. In collaboration with Oak Ridge National Lab, scattering of neutrons was additionally used to verify the magnetism. The result is an important step in the development of electrically-tunable magnets, with implications for next-generation computing paradigms.
Principal Investigators:
Chris Leighton (IRG-1)
Dan Frisbie (IRG-1)
About the figure: X. Ren, C.D. Frisbie, C. Leighton, J. Phys. Chem. Lett. 9, 4828 (2018)
Due to low cost, ease of fabrication, and flexibility, organic semiconductors hold great promise for technology. Full understanding of their electronic properties remains elusive, however. One example is the widely observed “conductance peak” in organic semiconductor electrolyte gate transistors. In these devices, a central focus of IRG-1 research, the conductance first increases with increasing gate voltage, before unexpectedly decreasing at the largest voltages, limiting performance. Using the prototype material rubrene, recent work in IRG-1 has gained important insight into the origin of this phenomenon. Specifically, detailed temperature-dependent measurements revealed not only that the temperature dependence of the conductance is highly anomalous, but also that it depends on the rate of cooling. The significance is that this is a hallmark of a phenomenon known as charge ordering, where electrons order into patterns. Charge ordering has not been previously considered in these devices, but provides an intriguing potential explanation for the origin of this limiting effect.
Principal Investigators:
Christy Haynes (IRG-2)
Uwe Kortshagen (IRG-2)
Zhi, B.; Mishra, S.; Hudson-smith, N.V.; Kortshagen, U.R.; Haynes, C.L. Toxicity Evaluation of Boron- and Phosphorus-Doped Silicon Nanocrystals toward Shewanella oneidensis MR-1. ACS Applied Nano Materials, 2018, 1 (9), 4884-4893.
Pramanik, S.; Hill, S.K.; Zhi, B.; Hudson-Smith, N.V.; Wu, J.J.; White, J.N.; McIntire, E.A.; Kondeti, V.S.K.; Lee, A.L.; Bruggeman, P.J.; Kortshagen, U.R.; Haynes, C.L. Comparative toxicity assessment of novel Si quantum dots and their traditional Cd-based counterparts using bacteria models Shewanella oneidensis and Bacillus subtilis. Environmental Science: Nano, 2018, 5 (8), 1890-1901.
Silicon and germanium nanocrystals are attractive materials for many electronic and optoelectronic applications. As part of its research portfolio, IRG-2 not only explores the physical properties of gas-phase produced nanocrystal materials, but also assesses their environmental impact. In a series of studies, IRG-2 researchers found that, different from many heavy metal-containing colloidally produced nanocrystals, pure (undoped) silicon and germanium nanocrystals present a negligible toxic risk to the environmentally important bacteria Shewanella oneidensis and Bacillus subtilis. However, doping with common elements such as boron and phosphorous can elicit a negative impact. These effects have been attributed to reactive oxygen species that are produced by the doped nanocrystals and negatively impact the viability of environmental bacteria. The studies regarding silicon nanocrystals have been published in Environmental Science: Nano and ACS Applied Nanomaterials.
Principal Investigators:
Frank Bates (IRG-3)
Cari Dutcher (IRG-3)
Timothy Lodge (IRG-3)
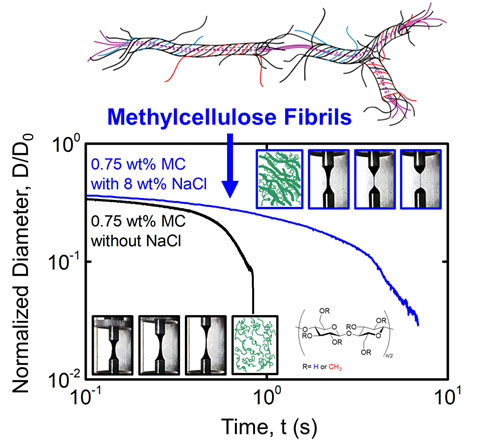
About the figure: S. Morozova, P. W. Schmidt, A. Metaxas, F. S. Bates, T. Lodge, C. S. Dutcher, ACS Macro Letters, 7, 347-352 (2018)
Understanding the behavior of polymer solutions in an extensional flow field is essential for a wide variety of processes including extrusion, spraying, and coating. Recently, a variety of methods have been developed to accurately characterize extensional properties of polymer solutions, such as capillary breakup extensional rheometry (CaBER). CaBER was used to measure the extensional properties of aqueous methylcellulose (MC) solutions with and without salt (NaCl). Upon the addition of salt at room temperature, MC chains form MC fibrils. The fibrillar network increases the extensional relaxation time and apparent extensional viscosity of the solution. Varying the MC and salt concentrations accesses a regime of stable extensional flows, which have never before been characterized for aqueous MC solutions. These measurements inform the polymer’s properties when exposed to industrial processes such as fiber spinning and extrusion.
Principal Investigators:
Sungyon Lee (Seed)
Xiang Cheng (Seed)
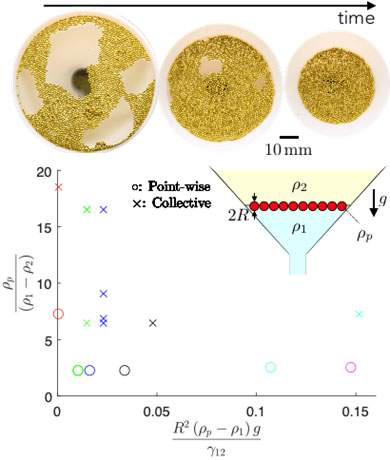
About the figure: B. Druecke, X. Cheng and S. Lee, In Preparation
Cereals floating on milk often clump together to form larger pieces. Known as the “Cheerios effect”, this commonplace phenomenon is caused by the interfacial tension between two fluids (e.g. milk and air) that mediates the interaction between floating objects. In recent years, scientists and engineers have harnessed the same physical mechanism to form synthetic materials from floating particles on a fluid-fluid interface, referred to as “particle self-assembly.” We currently investigate how the dynamics of the particle assembly may change if the fluid-fluid interface is undergoing expansion or compression. Our experimental set-up consists of a single layer of particles between two fluids inside a funnel; we visualize particle interactions as the fluid on the bottom is either drained from or injected into the funnel, causing the interface to shrink or to expand. Our results demonstrate that particles respond to interfacial compression in two different modes: by creating wrinkles collectively or by falling off the interface into the bottom fluid one by one.
Principal Investigators:
Sang-Hyun Oh (Seed)
Tony Low (Seed)
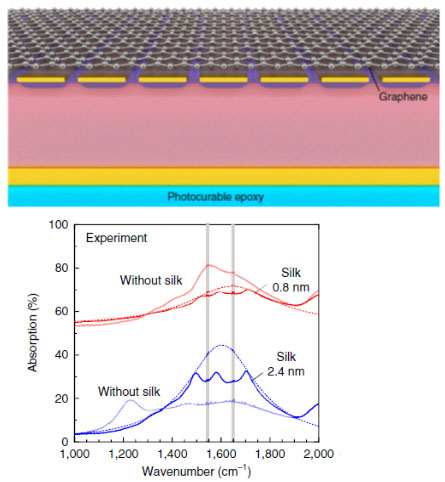
Sensing molecular vibrational modes has traditionally been very difficult to achieve at the sub-monolayer detection level. Fundamental to this challenge is the large mismatch between the mid-infrared wavelength and the atomically thin molecular layer. Graphene plasmons, which reside in the mid-infrared frequency, allow the surface confinement of electromagnetic energy down to within 50 nm. Our goal is to further shrink that down to just a few nanometers. Through novel design of the graphene-metal metasurface, we engineered a platform where mid-infrared light is squeezed into a nanometer-scale gap. This is achieved through a unique fabrication technique combining template stripping and atomic layer deposition (top figure), and the experimental platform is utilized to performed infrared fingerprinting of sub-monolayer-thick silk proteins (lower figure) and silica films.
Education and Outreach Highlight

About the figure: Top: MRSEC graduate students Beth Dewing and Athena Metaxas with visiting AI students in the UMN Valspar Materials Lab. Bottom: Prof. Vlad Pribiag discussing low temperature physics in the Physics and Nanotechnology building
In July of 2018, the inaugural American Indian Summer Institute was held at the University of Minnesota. The MRSEC, in collaboration with the American Indian studies department, held a one-week, residential camp for American Indian high school students in Minnesota. Led by MRSEC faculty and graduate students, 15 students participated in hands on laboratory activities designed to introduce them to a variety of topics in Materials Science and Engineering and the educational and research opportunities available to undergraduates at UMN. In addition to the STEM activities, students also had sessions in native languages (Dakota, Ojibwe), traditional medicine, and other culturally significant topics. Students participated in daily college-prep activities in writing, test taking (ACT/SAT), and admissions/application prep. The overall goal of the program is to increase the number of Native students enrolling at UMN and specifically in STEM disciplines.
Principal Investigators:
Xiaojia Wang (IRG-2)
Chris Leighton (IRG-1)
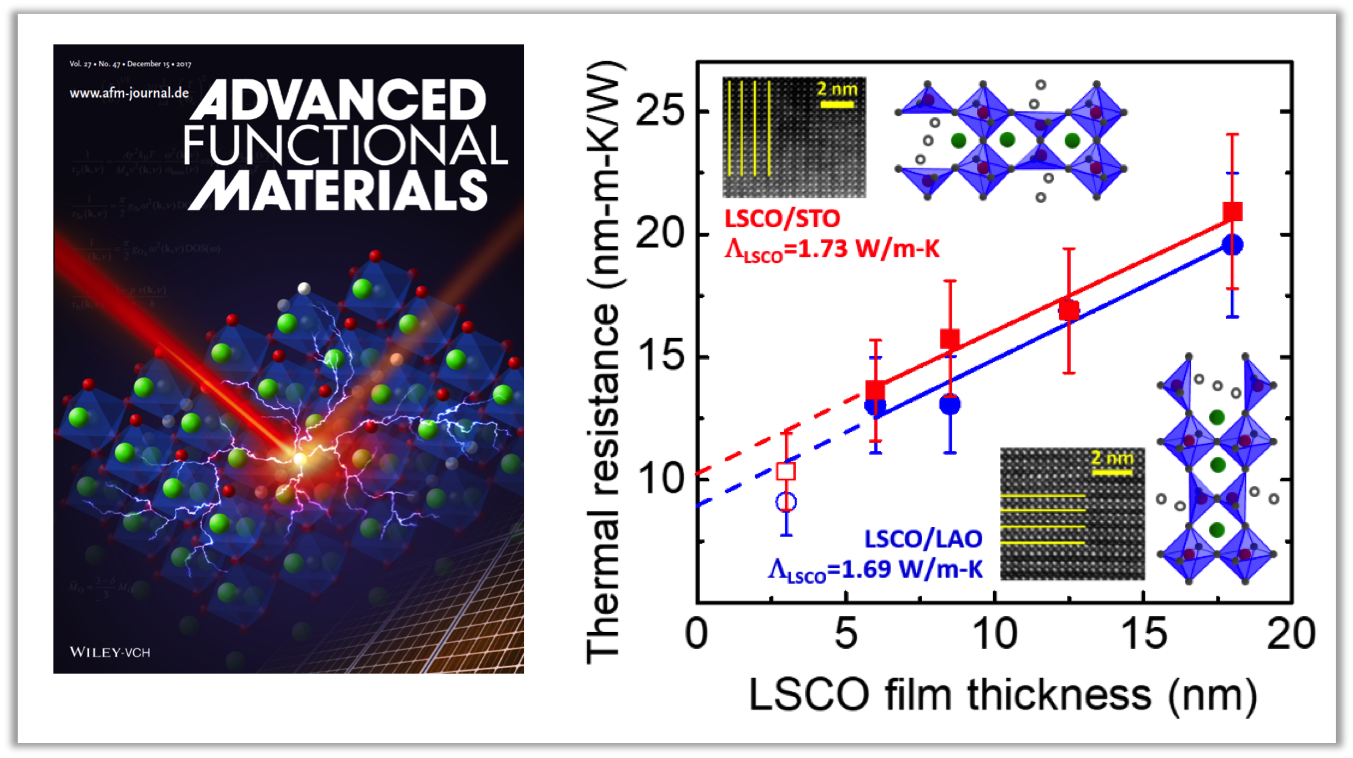
About the figure: from X.W. Wu, J. Walter, T.L. Feng, J. Zhu, H. Zheng, J. Mitchell, N. Biškup, M. Varela, X.L. Ruan, C. Leighton, and X.J. Wang, Adv. Funct. Mater.. 27, 1704233 (2017). DOI: 10.1002/adfm.201704233
Precise control over defects in materials is often a highly effective means to control properties and function. In oxide materials, which are the focus of enormous current attention for many existing and proposed applications, defects known as oxygen vacancies often play the key role. These vacancies, simply missing oxygen atoms in the structure, can have a significant impact on properties. Recent work by investigators in IRG-1 has shown that in a specific type of oxide (perovskite cobaltite films) oxygen vacancies can not only be created, but also controlled to form ordered patterns, in turn controlling electronic and magnetic properties. In this work, state-of-the-art capabilities in IRG-2 with measurement of thermal conductivity in nanoscale materials was used to measure the impact of this defect formation and ordering on the flow of heat. Surprisingly, the thermal conductivity in such materials was found to be extremely low, comparable to glasses, some of the best thermal insulators known. The finding that thermal insulation of this type can be obtained even in highly ordered crystalline materials has significant potential implications in heat management in devices.
Principal Investigators:
Chris Leighton (IRG-1)
Martin Greven (IRG-1)
Turan Birol (IRG-1)
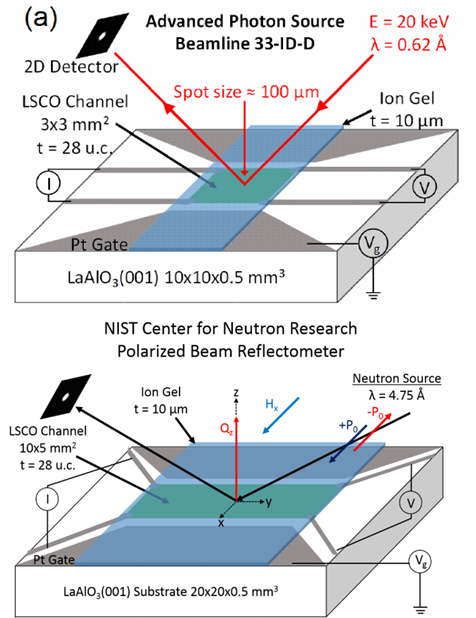
About the figure: from J. Walter, G. Yu, B. Yu, A. Grutter, B. Kirby, J. Borchers, Z. Zhang, H. Zhou, T. Birol, M. Greven, C. Leighton, Phys. Rev. Mater. 1, 071403(R) (2017). DOI: 10.1103/PhysRevMaterials.1.071403
One of the major concepts under exploration in IRG-1 in the U. of MN MRSEC is “electrolyte gating”, a highly promising potential means to electrically control materials properties. One of the difficulties in obtaining full understanding of the operating mechanisms in electrolyte-gated devices is characterizing them “in operando”, i.e., as they are actually functioning. Recent work in the IRG has made a breakthrough in this regard by establishing not one, but two, effective means to achieve this. Large-scale national facilities have been used to probe working devices using high energy X-rays and polarized neutrons, providing vital information on mechanisms. In particular, the devices are found to operate completely differently for different signs of the electrical voltage, a major advance in terms of resolving the ongoing debate surrounding mechanisms in this exciting technology.
Principal Investigators:
Uwe Kortshagen (IRG-2)
Eray Aydil (IRG-2)
Boris Shklovskii (IRG-2)
Lorraine Francis (IRG-2)
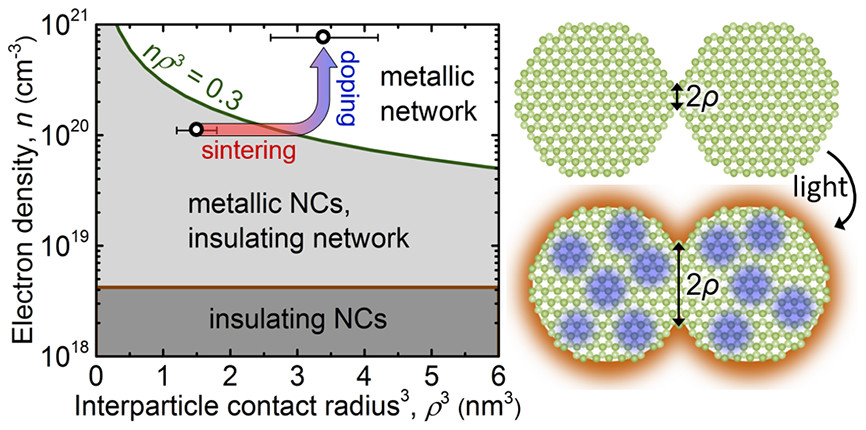
About the figure: We control our position on the electronic phase diagram of ZnO nanocrystal networks by sintering (increasing ρ) and doping (increasing n). Both sintering and doping are achieved with intense pulsed light (IPL). B. Greenberg, Z. Robinson, K.V. Reich, C. Gorynski, B. Voigt, L. Francis, B. I. Shklovskii, E. S. Aydil, M. U. R. Kortshagen, Nano Lett. 17, 4634 (2017). 10.1021/acs.nanolett.7b01078
Electrically conductive networks of semiconductor nanocrystals (NCs) are needed for a variety of optoelectronic devices. The small size of the nanocrystals gives rise to valuable new properties such as tunable light absorption. However, most semiconductor NC networks are poor conductors of electricity (“insulators”). In this work we explore the possibility of transforming such a network into a good conductor (“metal”) by applying intense pulsed light (IPL). We find that IPL improves conductivity in two ways: it increases both the interparticle contact area as well as the number of free electrons. Tuning both parameters, we observe electrical behavior indicative of close proximity to the insulator-metal transition. This allows us to evaluate the insulator-metal transition criterion predicted by Kortshagen and Shklovskii (Nature Materials., 2016) and to formulate strategies for obtaining truly metallic behavior. This study was reported in Nano Letters.
Principal Investigators:
Xiaojia Wang (IRG-2)
Stephen Campbell (IRG-2)
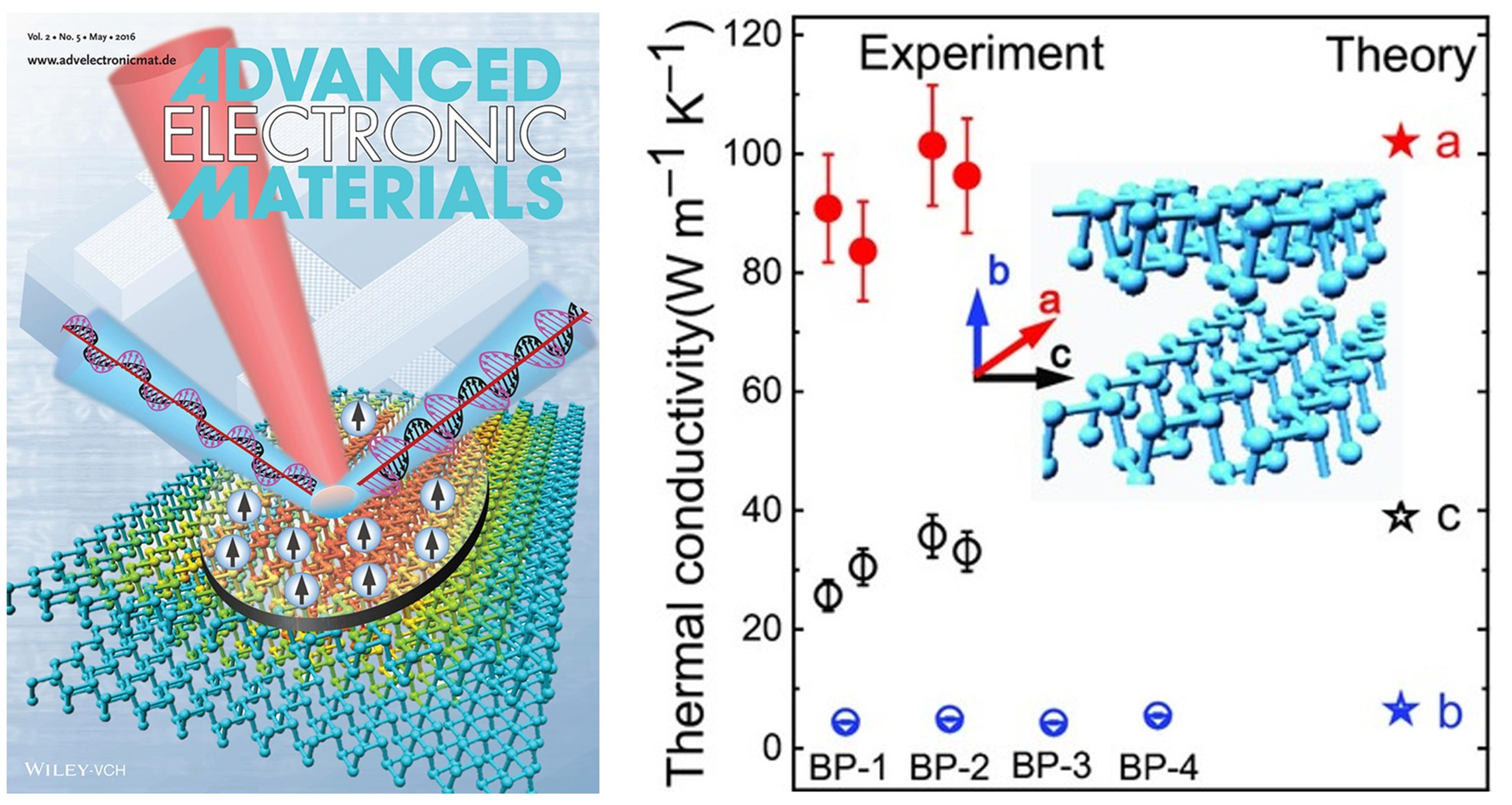
About the figure: J. Zhu, H. Park, J.Y. Chen, X.K. Gu, H. Zhang, S. Karthikeyan, N. Wendel, S. Campbell, M. Dawber, D. Xu, M. Li, J.P. Wang, R.G. Yang, and X.J. Wang, Adv. Electron. Mater. 2, 1600040 (2016). DOI: 10.1002/aelm.201600040
Black phosphorus (BP) has emerged as a direct-bandgap semiconducting material with great application potentials in electronics, photonics, and energy conversion. Experimental characterization of the anisotropic thermal properties of BP that are micrometers in size is extremely challenging. One difficulty comes from the challenge in synthesizing large-size high-quality BP crystals. Another impediment stems from the lack of reliable and accurate experimental techniques to measure the anisotropic thermal properties of thin-film samples. In this work, we synthesize large-size high-quality BP crystals and employ a novel technique of time-resolved magneto-optical Kerr effect (TR-MOKE) to study their intrinsic anisotropic thermal conductivities along three primary crystalline orientations. This work reveals the origins of the anisotropic thermal transport in BP, as demonstrated by the excellent agreement between measurement results and first-principles calculations. This collaborative work between two PIs of IRG-2: Xiaojia Wang (ME) and Stephen Campbell (ECE) has been featured as the cover image of Advanced Electronic Materials.
Principal Investigators:
Tim Lodge (IRG-3)
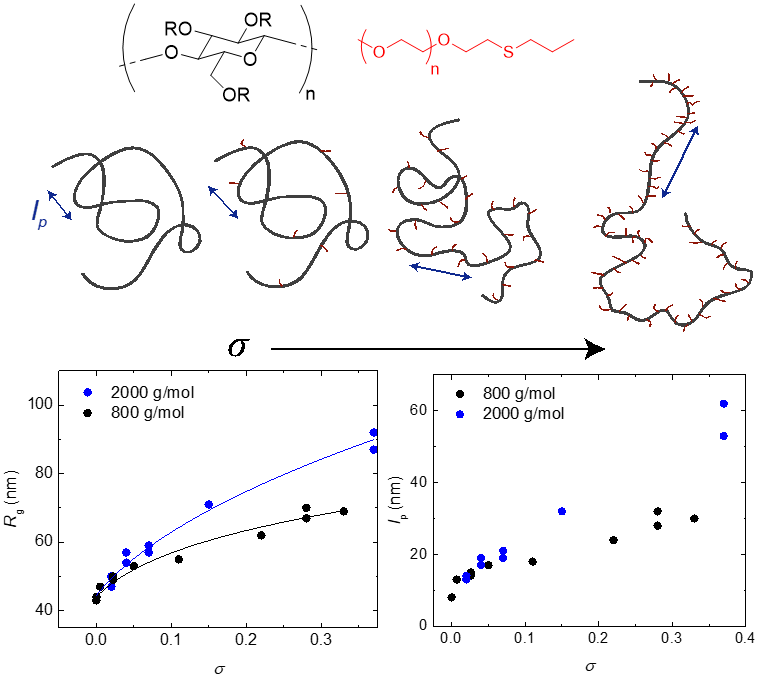
About the figure: S. Morozova and T. P. Lodge. ACS Macro Letters. 2017, 6, 1274. DOI: 10.1021/acsmacrolett.7b00776
Methyl cellulose (MC) is a widely used plant derived polymer with a unique property – at high temperatures in aqueous solutions the chains self assemble into fibrillar networks. The radius of the fibrils is remarkably uniform and is likely set by the persistence length (lp) of the MC chains. To test this hypothesis, we have systematically grafted 800 and 2000 g/mol poly(ethylene glycol) chains onto the backbone of MC using thiol-ene click chemistry, with grafting densities (σ) from 0.07 –- 0.33 (PEG molecules per anhydroglucose repeat unit). Light scattering reveals that at room temperature grafts effectively stiffen the chain, increasing both the overall radius and the persistence length of the MC backbone. In the most extreme case of highly grafted 2000 g/mol PEG, lp increases by an order of magnitude. This finding agrees with theoretical predictions exceptionally well, and provides a systematic approach to increasing MC chain stiffness.
Principal Investigators:
Ilja Siepmann (IRG-3)
Tim Lodge (IRG-3)
Marc Hillmyer (IRG-3)
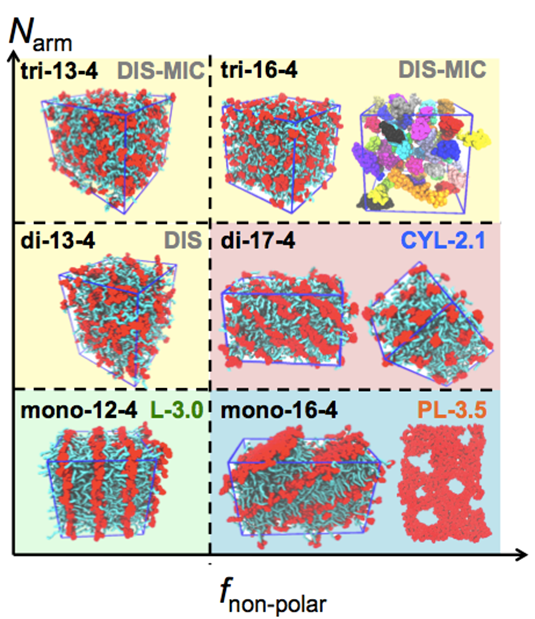
About the figure: Simulation snapshots of meso-phases formed by H(CHOH)x(CH2)yH oligomers (polar in red and nonpolar in cyan) including Lamellae, Perforated Lamellae, Cylinders, and Disordered Micelles. Q. P. Chen, L. Barreda, L. E. Oquendo, M. A. Hillmyer,T. P. Lodge, J. I. Siepmann, ACS Nano. 12, 4351-4361 (2018). DOI: 10.1021/acsnano.7b09122
The ability to precisely predict how molecular structure influences the microstructure of polymeric materials is the key towards the custom tailoring of desirable materials properties. Molecular dynamics simulations with atomistic level models were performed to design “high-χ” block oligomers that can self-assemble into 1-5 nm domains for next generation microelectronics applications. Simulations show that the microstructures formed by these oligomers can be tuned by varying the molecular weight and the chain architecture. The results are being used to guide the synthesis of block oligomers for desired microstructural morphologies and domain periods. In particular, these results open a path to significantly smaller features than previously obtained by block polymer self-assembly.
Principal Investigators:
Tony Low (Seed)
Sang-Hyun Oh (Seed)
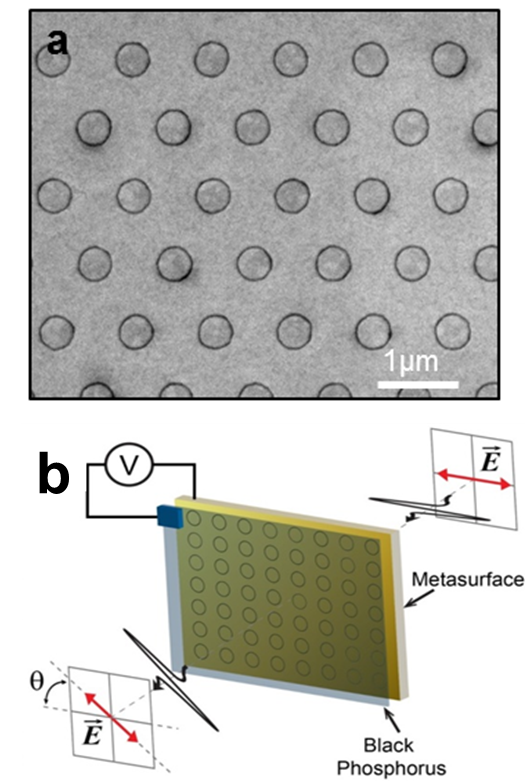
About the figure:I.-H. Lee, L. Martin-Moreno, D. A. Mohr, K. Khaliji, T. Low, S.-H. Oh. ACS Photonics. 2018. DOI: 10.1021/acsphotonics.8b00062. Physical Review B, 2017, 95 (20), 201401. DOI: 10.1103/PhysRevB.95.201401
Everyday optical waveplates used for polarization control of scattered light rely on bulky birefringent materials. Our goal is to realize such functionality on an atomic scale with the effect also being electrically tunable. To achieve this, we are exploring the use of plasmons in a highly anisotropic two-dimensional material called “black phosphorus” to realize an extreme form of birefringence. We predict that metasurfaces exploiting such anisotropic plasmons could lead to arbitrary control of the polarization of reflected light, including polarization rotation and conversion in the mid-infrared regime, and could have applications in displays and imagers. Through unique atomic layer deposition techniques (Figure a), we are building an experimental platform for realizing polarization control with only atomic thin materials (Figure b).
Education and Outreach Highlight

About the figure: Top: Group Photo of 2017 workshop participants; Bottom: RET participant and workshop instructor, Cassie Knutson (center) demonstrating workshop lab activities.
The Green and Sustainable Chemistry workshop was created in 2017 to share the research and curriculum developed by UMN MRSEC RET participants and help ensure RET activities reach a larger student audience. In its inaugural year, 20 high school teachers completed the three-day workshop. During the workshop, teachers learned three separate lab activities designed to incorporate materials science and engineering concepts into the high school chemistry classroom. All three activities meet Next Generation Science Standards in engineering. Teachers who completed the workshop were provided with all materials necessary to implement the new lab activities in their classrooms – removing a significant barrier to the introduction of new class activities.
Principal Investigators:
Ilja Siepmann (IRG-3)
Timothy Lodge (IRG-3)
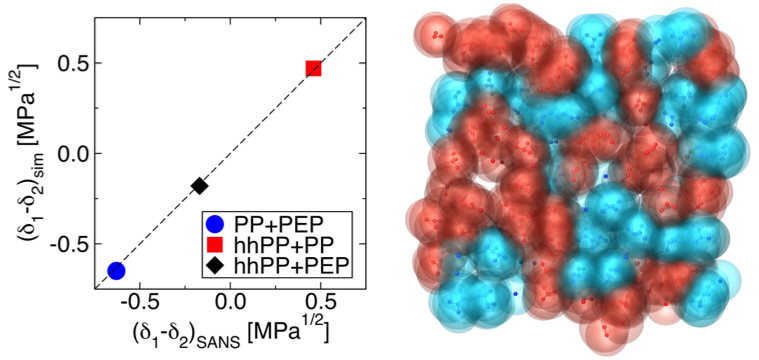
About the figure: (Left) Comparison of solubility parameter differences determined from scattering experiments and calculated from molecular simulations: polypropylene (PP), head-to-head polypropylene (hhPP), and poly(ethylene-alt-propylene) (PEP). (Right) Snapshot of PP (red) and hhPP (blue) blend.
The versatility of polymeric materials is inherent in their rich and varied molecular building blocks and architectures. The ability to quantitatively model polymer systems on the molecular level provides a route to engineer polymeric materials with desired properties. In this work, we utilize Monte Carlo simulations and transferable force fields to study the thermodynamic and structural properties of some olefin oligomers. It is shown that extrapolation of data for short oligomers can reproduce important thermodynamic properties of the polymers. In addition, the Flory−Huggins χ parameters, a measure of the unlike bead-bead interactions, are calculated directly from simulations of binary mixtures. The binary oligomer blend of propylene isomers is found to exhibit stabilized irregular mixing behavior, in agreement with experiments for polymers. Our results identify molecular simulations as a promising approach to predict and understand the polymer phase behavior.
Principal Investigators:
Timothy Lodge (IRG-3)
Kevin Dorfman (IRG-3)
David Morse (IRG-3)
Theresa Reineke (IRG-3)
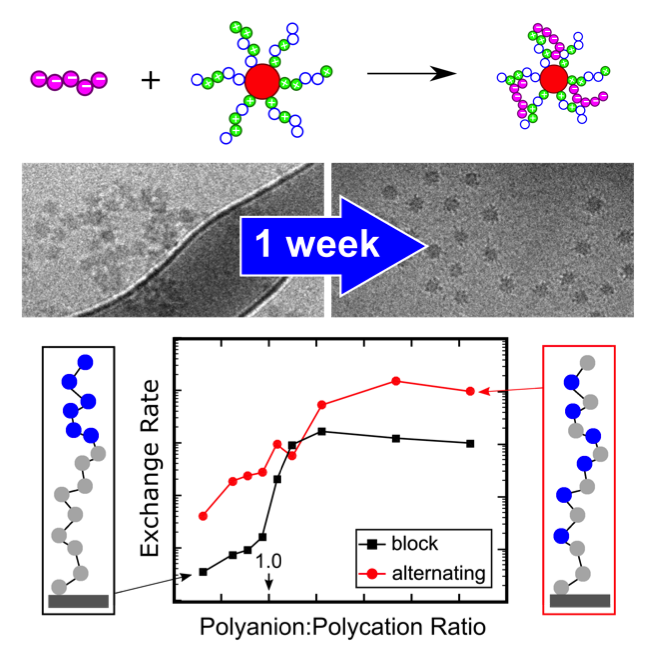
A significant challenge facing the development of polyelectrolyte complexes for biomedical and materials applications is kinetic trapping of the complexes far from their equilibrium states. Recent work from IRG-3 demonstrates that controlling the charge density of the polyelectrolyte chains can help promote rapid equilibration of these complexes. Through experiments on complexes of polyelectrolyte micelles, researchers in IRG-3 showed that incorporation of neutral, uncharged monomers facilitates rapid rearrangement from insoluble multi-micelle aggregates to soluble single-micelle complexes. Molecular dynamics simulations on model polymer brushes demonstrated that this results from the energy required to break ion-ion contacts in polymers with alternating charged and neutral units being lower than in polymers with many adjacent charges. These results provide critical new information for applications requiring rapid equilibration followed by long-term stability.
Education, Outreach, and Diversity Highlight

In 2016, the MRSEC broadened the reach of its popular Materials Week program by presenting hands-on demonstrations and lab activities for 30 Middle School girls participating in the Eureka! program. Eureka! is a partnership between UMN and the MPLS YWCA to encourage participation in STEM by providing opportunities to girls beginning in 7th grade and supporting them continuously through high school graduation. The MRSEC Materials Week program began as a weeklong summer camp for high school students but has since grown to include students as young as 7th grade. The library of activities developed for the program are now used is several MRSEC Education and Outreach programs and presented to nearly 300 middle and high school students each year.
Principal Investigators:
Uwe Kortshagen, (IRG-2)
Andre Mkhoyan (IRG-2)
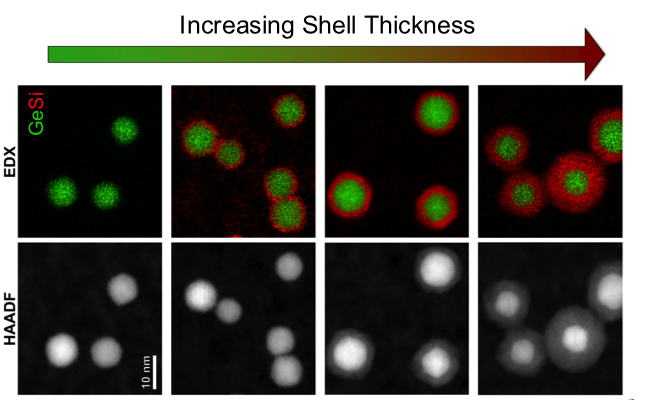
About the figure: Core/shell nanocrystals with increasing shell thickness as viewed by Scanning/Tunneling Electron Microscopy (STEM) (bottom) and Energy Dispersive X-ray Spectroscopy elemental (top). Shells of increasing thickness are grown by increasing the concentration of silane (SiH4) in the shell gas feed.
Nanoparticles, consisting of only a few hundred to a few thousand atoms, are of interest as building blocks for bottom-up processing or next generation electronics. Because they are so small, a significant fraction of the atoms that make up these materials are found at the surface. Consequently, these surface atoms are extremely important in determining the properties and stability of the nanomaterial. In this work, the researchers demonstrated the ability to grow nanoparticles with a shell of a different material, all in the gas-phase using a plasma. This process allowed them to make core/shell nanocrystals that required high temperatures for synthesis that would be difficult or impossible to achieve in solution. They demonstrated the process by synthesizing germanium nanocrystals (<10 nm) with a silicon shell produced using a novel nonthermal plasma reactor design (K. Hunter, U. Kortshagen; Department of Mechanical Engineering). In collaboration with colleagues in the Department of Chemical Engineering and Material Science (J. Held, A. Mkhoyan), the core/shell structure of these nanocrystals was confirmed by scanning tunneling electron microscopy (STEM). Through careful analysis of these STEM images, the researchers were able to correlate the change in the properties of these particles with increasing shell thickness. The ability to use gas-phase processes to create nanoparticles with a core/shell structure opens up the possibility to produce nanomaterials that have so far been inaccessible by other approached. This study was reported in the journal ACS Applied Materials and Interfaces.
Principal Investigators:
Rafael Fernandes (IRG-1)
Chris Leighton (IRG-1)
Boris Shklovskii (IRG-2)
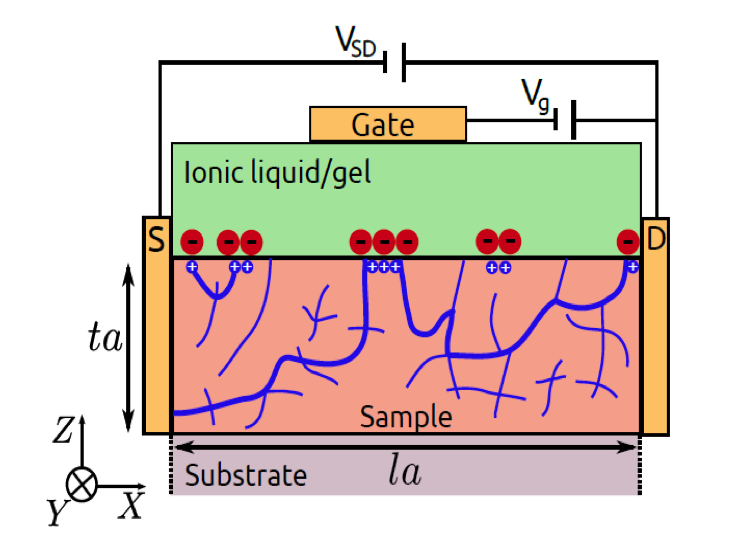
A new concept being developed in MRSEC IRG-1 at UMN allows for external electrical control over the electronic and magnetic properties of materials. This is based on the use of electrolytes in transistor devices, using electrical voltage to inject electrons into material surfaces, thereby controlling their properties. In this work, researchers in IRGs 1 & 2 have studied this problem theoretically, making predictions about the surface electron density required to induce transitions from insulating to metallic, and from non-magnetic to magnetic. The latter proceeds by connecting together (“percolating”) isolated magnetic clusters, forming a full long-range magnet. This work predicts that even though the electrons are added only at the surface, the percolated magnetism can extend much deeper into the material than expected, an exciting prediction that is now being tested in experiments.
Principal Investigators:
Dan Frisbie (IRG-1)
Chris Leighton (IRG-1)
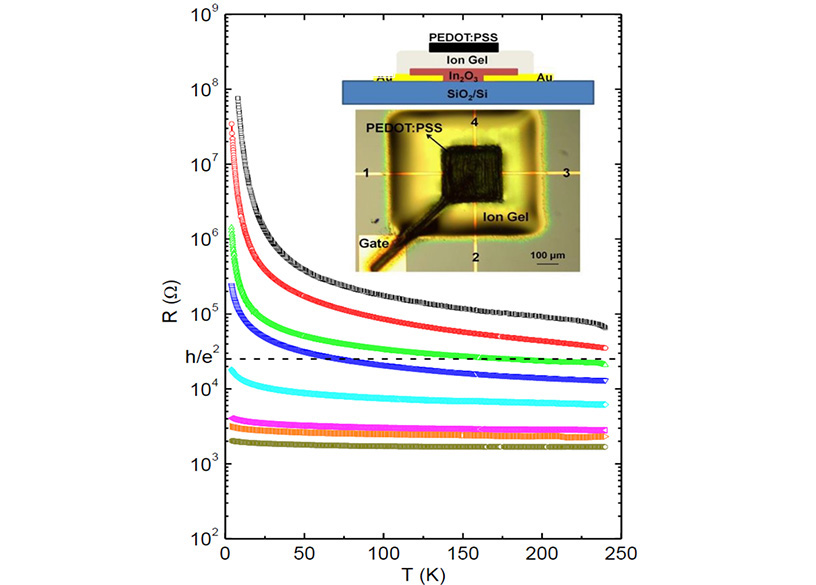
Transistors, the building blocks of all computer technologies, are currently based on semi-conductors such as silicon, manufactured using energy-intensive processes. Materials that can be processed into electronic devices using cheaper and less energy-intensive methods are of high interest for a number of applications. In work recently performed in IRG-1, UMN MRSEC researchers have demonstrated landmark performance in transistors based on the widely studied transparent semiconductor indium oxide, fabricated via solution processing. Solution processing is a low temperature, low cost approach (in this case essentially a form of inkjet printing), but was shown here to be capable, in conjunction with cutting-edge electrolyte dielectrics, of voltage-induced metallic behavior at interfaces. This metallic conductivity is important, as it maximizes current output, improving device performance and applicability.
Principal Investigators:
K. Andre Mkhoyan (IRG-2)
Renata Wentzcovitch (IRG-1)
Bharat Jalan (IRG-1)

Defects, essentially locations in a crystal where the perfect arrangement of atoms is disturbed, are inherent in materials, and play a key role in their function. As they have been studied for so long, the discovery of new types of defects is rare. In recent work in IRGs 1 and 2 a completely new type of defect has been found in a class of complex oxide materials called perovskites, specifically the compound NdTiO3. This new defect is a “line” defect formed by a long local region of the crystal that rotates from the typical ordering pattern, as shown in the figure. This discovery was enabled by combining state-of-the-art synthesis methods with atomic resolution electron microscopy and complex computations, requiring collaboration between three MRSEC research groups with complementary expertise. One potential application lies in engineering such line defects to create atomic-scale “tunnels” for the flow of electrons or atoms.
Principal Investigator:
Boris Shklovskii (IRG-2)
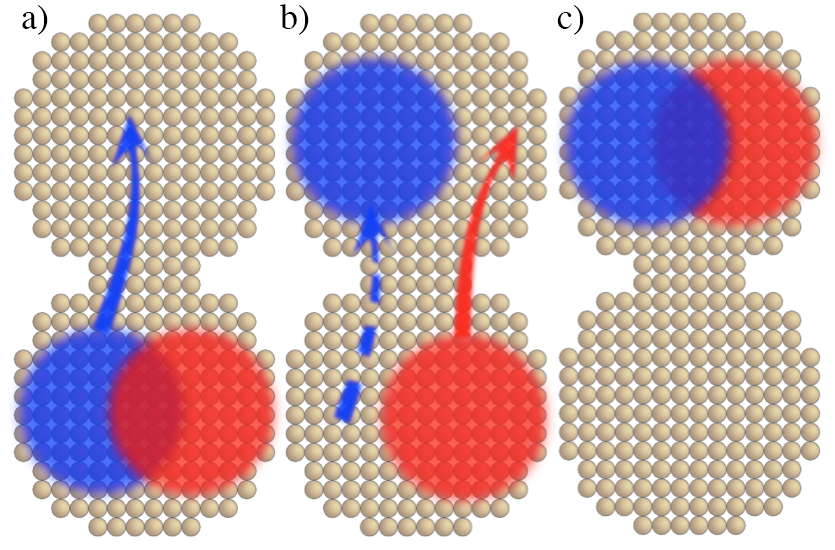
When a nanocrystal film absorbs a light quantum, its energy is stored in a nanocrystal exciton, which consists an electron and hole bound to each other by electrical attraction. The exciton hops between nanocrystals and, to release its electrical energy, should get converted into a free electron and hole before losing its energy. This is why it is important to increase the exciton hopping rate. The research team found a new fast mechanism of exciton hopping: first the electron moves from one nanocrystal to another and then the hole follows the electron. The research team showed that for touching semiconductor nanocrystals such hopping mechanism can be more effective than any other known mechanisms. This study was reported in ACS Nano.
Principal Investigator:
David Flannigan (seed)
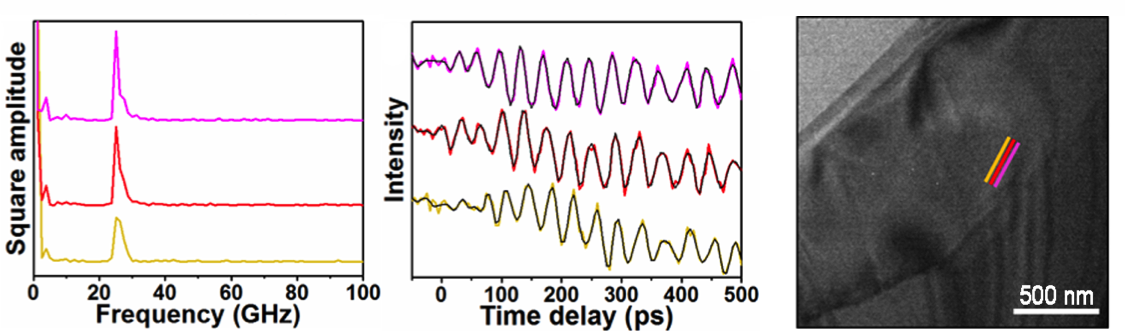
Energy in the form of heat impacts all technologies and plays a major role in the design and engineering of infrastructure. It is also the largest form of waste energy in critical applications, including power transmission and transportation. Scientists and engineers have spent decades researching how to control thermal energy at the atomic level in order to use it to do useful work and ultimately increase efficiencies and reduce the use of fossil fuels. However, no one has yet been able to directly image what thermal energy looks like or how it moves through materials in real time. This is because the basic length scales are billionths of a meter (nanometers) and the speeds can be many miles per second. Here, using a cutting-edge electron microscope, we were able to directly image the emergence and motion of exceedingly fast energy waves moving through semiconducting materials. Even more exciting was our observation that these energy waves interact with particular features in materials in ways that would have been impossible to determine with such certainty by any other means. These observations represent a breakthrough in our ability to study and understand how energy moves through materials and could potentially change the way we approach thermal-energy management.
Principal Investigators:
Bharat Jalan (IRG-1, Seed)
Vlad Pribiag (Seed)
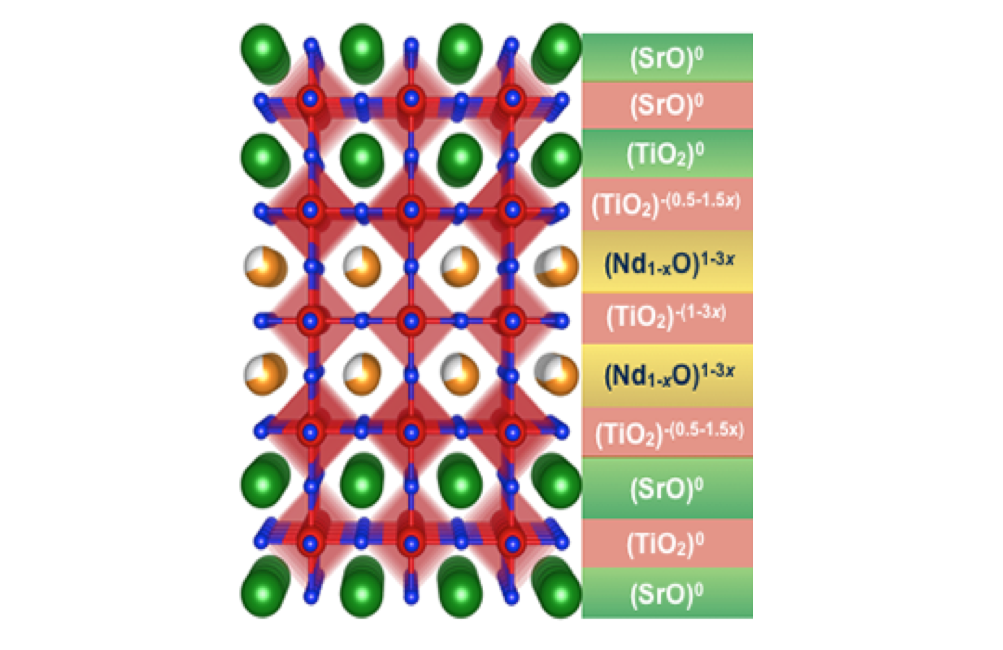
An important recent advance in the materials science of metal oxides is the discovery that interfaces between intrinsically non-conductive complex oxide materials can exhibit conductive behavior. This, and other advances, have led to the concept that “oxide electronics” could be developed, with functionality not possible in current devices. In this work, IRG-1 researchers, along with a SEED researcher and collaborators at the Pacific Northwest National Laboratory, have identified, for the first time, both the source of the electrons that conduct, and the means to control their number. In essence the interfacial electrons are controlled remotely (“by their tail”) by tuning the composition away from the interface. This is a significant advance in thin-film engineering of oxides, in that properties are controlled at the level of the individual atoms that make up the materials. Some of the materials used are only a single atomic layer thick, yet their properties can still be controlled. This discovery has several intriguing implications, including the possibility of new electronic and photonic devices.
Principal Investigators:
Uwe Kortshagen (IRG-2)
Eray Aydil (IRG-2)
Andre Mkhoyan (IRG-2)
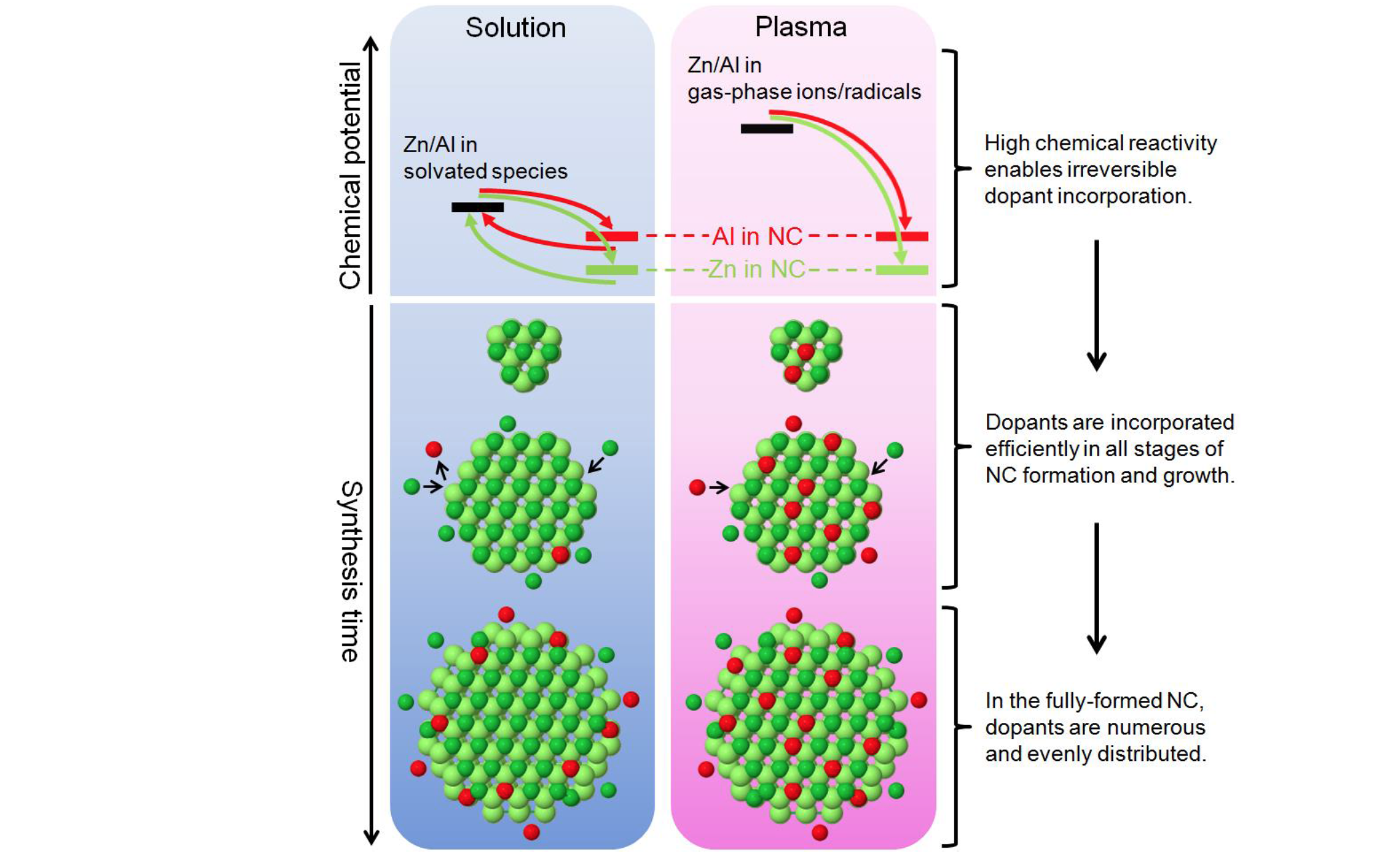
About the figure: Doping in solution vs. doping in plasma. Kortshagen, Aydil, Mkhoyan et al. Nano Lett. 2015, 15, 8162–8169.
Doping – one of the central challenges of nanocrystal engineering – is essential for controlling the optical and electronic properties of compound semiconductor nanocrystals. Conventionally, these materials are synthesized and doped by solution-based methods, but a significant obstacle is often encountered: dopants are excluded during the early stages of nanocrystal formation and growth, resulting in undoped central cores and low doping efficiencies. To address this problem, a team led by Professors Kortshagen, Aydil, and Mkhoyan developed a fundamentally different plasmabased process for synthesizing aluminum-doped zinc oxide nanocrystals, a prototypical material used in light-emitting diodes and solar cells. The key advantage of this approach was that the dopant Al atoms in the plasma were much more chemically reactive than their counterparts in solution-based synthesis. The team demonstrated that this high reactivity enabled irreversible dopant incorporation in all stages of nanocrystal growth, resulting in efficient and uniform doping throughout the nanocrystal cores.
Principal Investigator:
James Johns (seed)

The ability to control the flow of electrons in semiconductor electronics depends on the presence of junctions between materials with different electronic energy levels. Traditional techniques for creating interfaces in semiconductor materials do not work well for monolayer, or two dimensional, materials. Instead, junctions in 2D materials are typically formed by stacking one material on top of another, forming a weakly bound, vertical stack through which electrons can flow. Researchers sponsored by the SEED program have recently developed a new method for forming atomically abrupt junctions in 2D materials. In their work, they used chemical vapor deposition to grow monolayers of semiconductor MoS2. By introducing molecular hydrogen into the reaction, they were able to control the shape and cleanliness of their MoS2 flakes. They then performed a second reaction to deposit another monolayer semiconductor, WS2. They were able to chemically control whether the second material grew on top of MoS2, as a vertical stack, or whether it grew around the MoS2 forming a covalent junction similar to junctions found in traditional semiconductors. The figure below displays a chemical model of the ideally abrupt interface, an SEM image of the monolayer flakes, and a TEM image confirming the atomically sharp interface. The abrupt, lateral chemical junction creates an abrupt electrical, p-n junction which could be used for optoelectronics such as solar cells and LEDs.
Principal Investigator:
Marc Hillmyer (IRG-3)
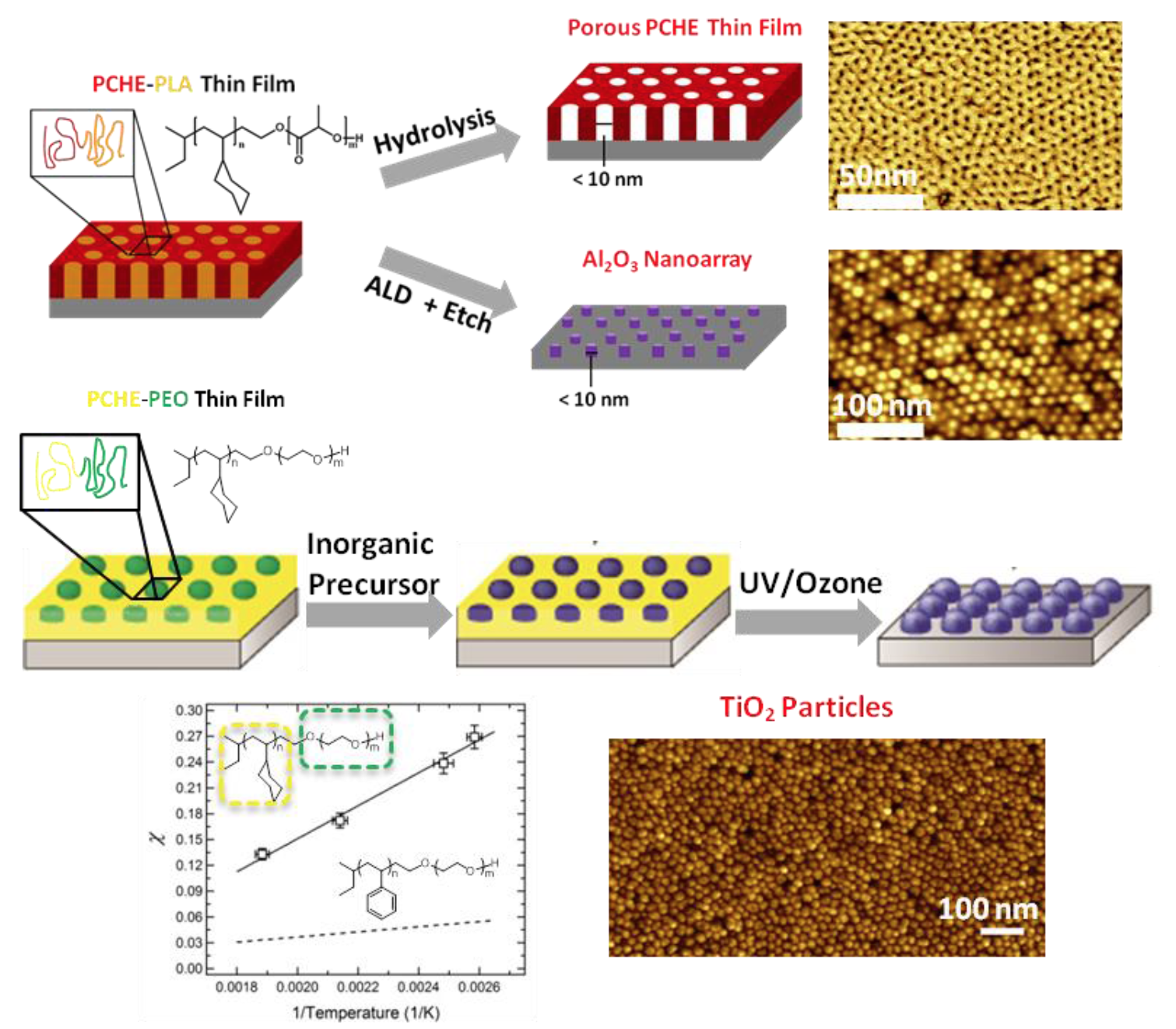
Block polymers can produce high density nanostructured arrays by the attractive “bottom-up” strategy of self-assembly. Strongly segregated block polymers with low degree of polymerization are needed to prepare ultrahigh density features for emerging applications in microelectronics and high density magnetic data storage. A series of novel poly(cyclohexylethylene)-blockpoly( lactide) (PCHE-PLA) and poly(cyclohexylethylene)-block-poly-(ethylene oxide) (PCHEPEO) polymers have been synthesized to achieve ultra-small nanostructured arrays with sub-10 nm domain sizes. Ordered block polymers thin films with ultra-small hexagonally packed cylinders oriented perpendicularly were prepared by spin-coating and subsequent solvent vapor annealing for use in three distinct templating strategies. Selective hydrolytic degradation of the PLA domains generated nanoporous PCHE templates with an average pore diameter of 5 ± 1 nm. Alternatively, an Al2O3 nanoarray from the PCHE-PLA template was produced on diverse substrates including silicon and gold with feature diameters less than 10 nm. In a third approach, selective inclusion of inorganic precursor within the PEO domain enabled the formation of inorganic oxide nanodots with exceptionally small feature sizes of 6 ± 1 nm.
Principal Investigators:
Uwe Kortshagen (IRG-2)
Boris Shklovskii (IRG-2)
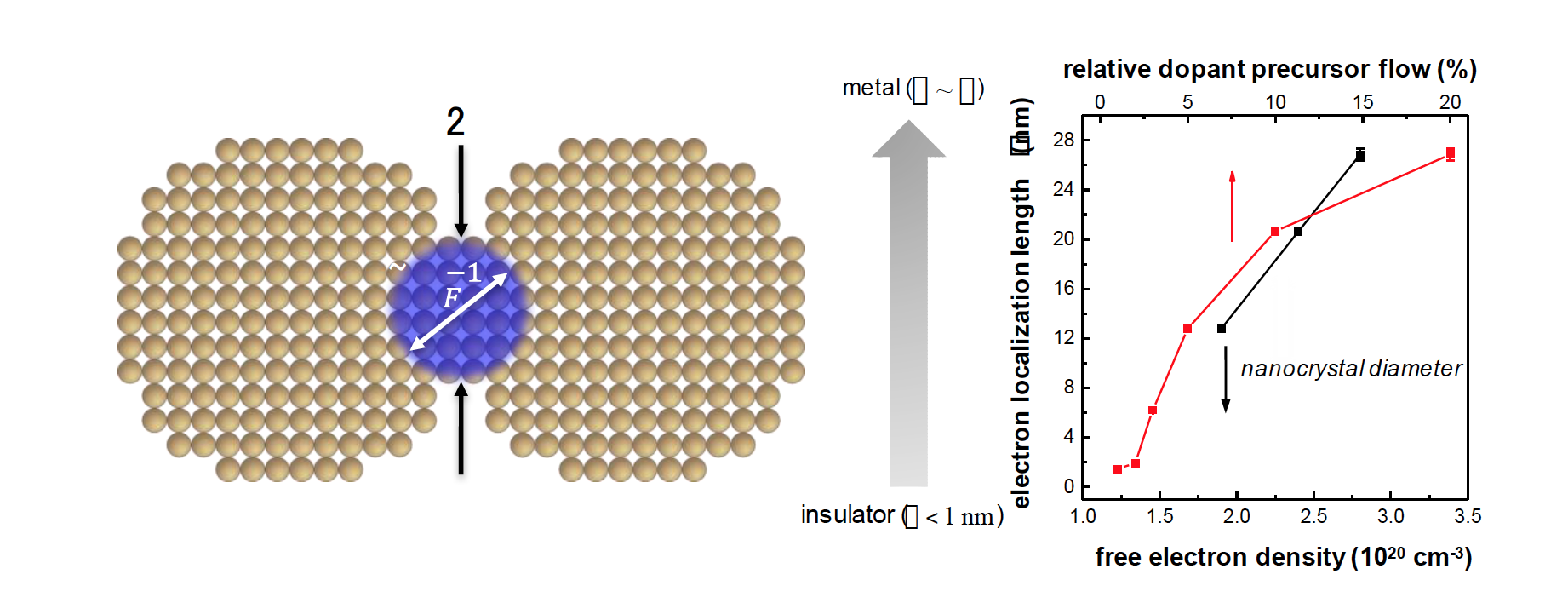
About the figure: The metal-to-insulator transition in nanocrystal films occurs when the spatial spread of the electron wave function, described by the inverse Fermi vector kF-1, allows the electron to pass through a contact with diameter 2𝜌. The Fermi vector kF increases with the free electron density and the doping level. Measurements of the electron localization length ξ in phosphorous-doped silicon nanocrystal films support the theory, as they indicate the approach to the metal-to-insulator transition.
The transition of a solid from an insulator to a metal as the number of free electrons in the material increases has been one of the central questions in semiconductor physics. In bulk semiconductors, this “metal-to-insulator transition” is described by the well-known Mott criterion. Understanding this transition in films of nanocrystals is of crucial importance to their future use in electronic devices such as light emitting diodes, solar cells, or transistors. An IRG-2 research team developed a new theory that relates the metal-to-insulator transition in films of doped nanocrystals to the “transparency” of the interface between nanocrystals for electrons. The theory predicts that the transition occurs under strikingly different conditions from those previously known for bulk semiconductors. In associated experimental studies of the electron conduction in phosphorousdoped silicon nanocrystal films, the team discovered a behavior that largely supports the predictions of the new theory. This study was reported in the journal Nature Materials.
Principal Investigators:
Frank Bates (IRG-3)
Lorraine Francis (IRG-2)
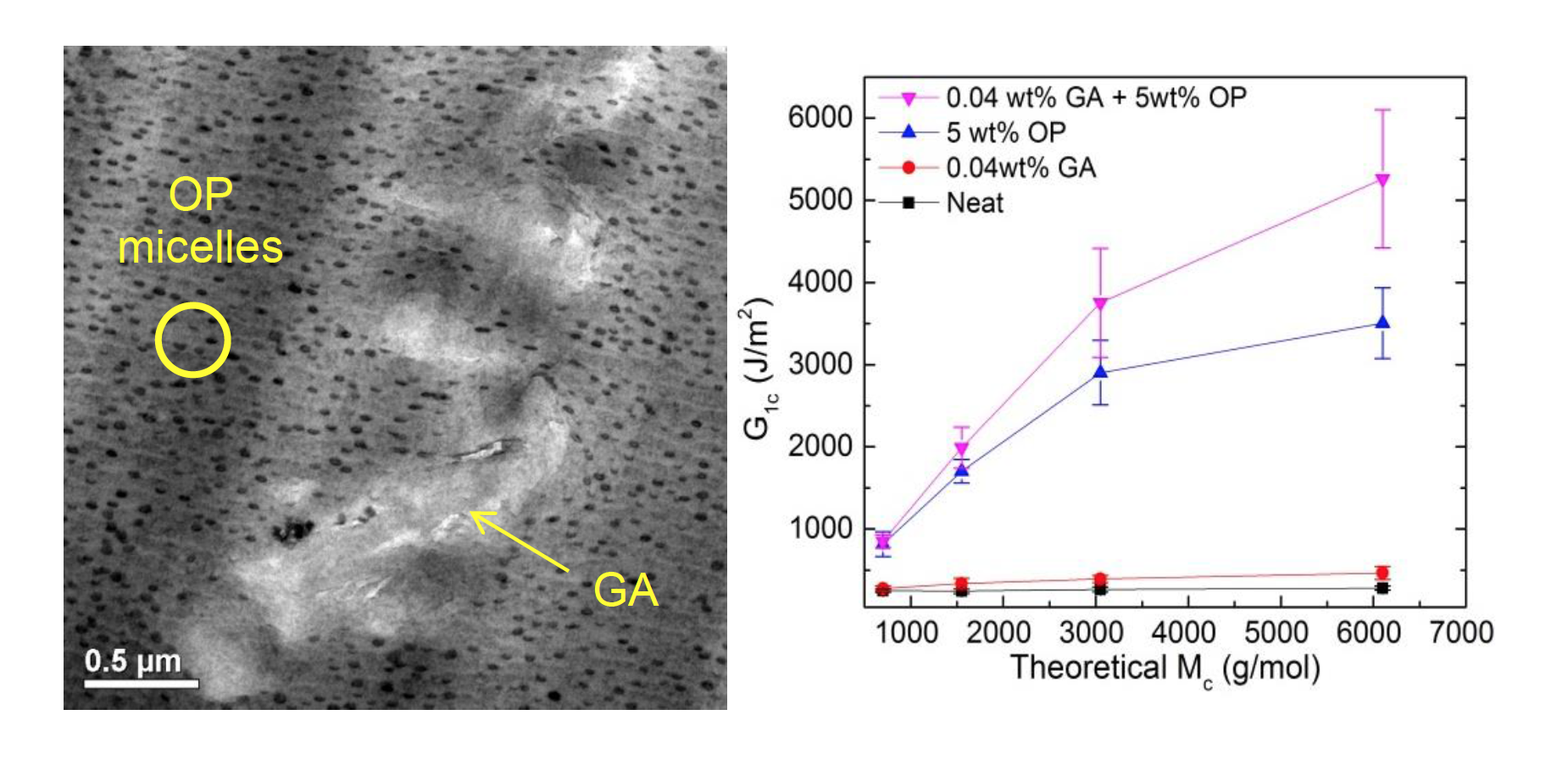
Glassy thermosets, such as epoxy, are brittle and lack the mechanical toughness needed for many applications. This work demonstrates that dispersing small amounts of nanoscale micelles of poly(ethylene-alt-propylene)-b-poly(ethyleneoxide) (OP) diblock copolymer (5 wt%) and amine modified graphene (GA) (0.04 wt%) to an epoxy results in an unprecedented 20-fold increase in the strain energy release rate (GIc), a measure of toughness. Remarkably, the improvement is multiplicative: graphene addition boosts the GIc of block copolymer modified epoxy by 1.8 times, the same increase noted for its addition to the neat epoxy material. Future work will focus on the underlying toughening mechanisms and the properties of composite coatings.
Principal Investigator:
Allen Goldman (IRG-1)
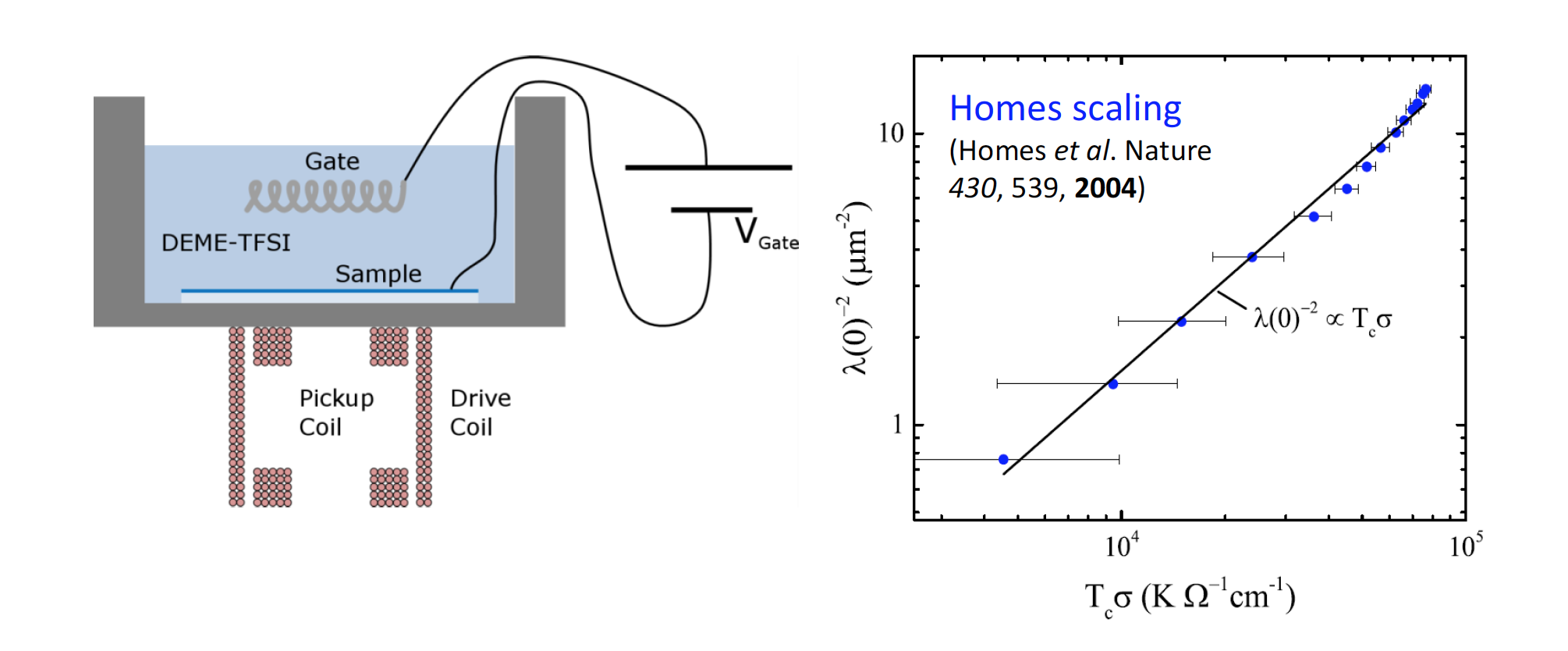
High temperature superconductivity remains one of the biggest challenges in condensed matter physics. One of the major materials issues with high temperature superconductors is the difficulty of chemically doping materials such as complex copper oxides (cuprates) over a wide range of charge carrier densities. Recently developed methods using ionic liquids in devices called electric double layer transistors provide an elegant potential solution to this problem as they enable doping not chemically, but rather by applying an external electrical voltage. In this recent work in IRG-1, investigators have shown that this ionic liquid approach can be used to establish a special scaling relation between the superconducting penetration depth and transition temperature in cuprates, known as Homes scaling. The ionic liquid gating approach is highly efficient compared to prior methods, the scaling being demonstrated in a single sample, tuned via an external parameter.
Principal Investigators:
Bharat Jalan (IRG-1)
Andre Mkhoyan (IRG-2)
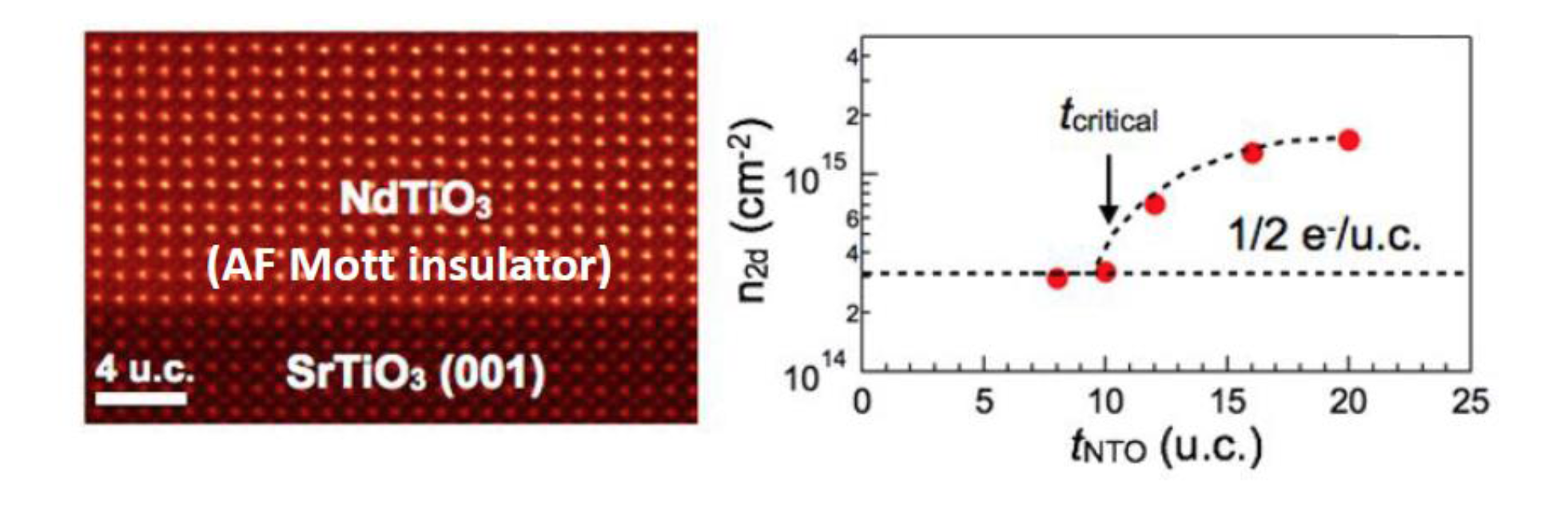
Complex oxides are extraordinarily functional materials, and are promising for next generation “oxide electronic” devices. One particularly attractive direction with such materials is the formation of two-dimensional (2D) conductive layers at the interfaces between insulating complex oxides. In work recently performed in IRG-1 an exciting development with these interfaces has been uncovered, arrived at by working in collaboration with researchers in IRG-2 and at the Pacific Northwestern National Lab. Specifically, the interface between the Mott insulator NdTiO3 and the band insulator SrTiO3 was shown to have an unusual energy band alignment that enables additional transfer of electrons from NdTiO3 to SrTiO3, thus creating an electron gas with almost ten times the electron density of standard interfaces. This phenomenon occurs at a critical thickness of the NdTiO3 layers, and is potentially externally controllable. The discovery has several intriguing implications, particularly for new photonic device concepts.
Education and Outreach Highlight

In 2015, the University of Minnesota MRSEC expanded its American Indian Outreach activities with the inaugural American Indian Visit Day. On November 7, 2015, 270 American Indian middle and high school students were invited to UMN for a day of activities to introduce opportunities in STEM available at UMN. The students participated in hands on activities presented by each of the three MRSEC IRGs exposing them to research in multiple science and engineering fields. Additionally, the students toured campus cultural offices and heard from American Indian student and alumni speakers. Students also participated in a college application workshop and were able submit applications to UMN on site at no cost.
Principal Investigators:
Bharat Jalan
Chris Leighton
Complex oxides such as perovskites are extraordinarily functional materials, and are promising for next generation “oxide electronic” devices. A weakness of these materials, however, is that they support high electron mobility at cryogenic temperatures, but this is difficult to translate to room temperature operation. BaSnO3, an emerging material with record room temperature mobility is thus of high current interest. In work performed in IRG-1, researchers have now demonstrated an effective and simple approach to doping this material, simply by annealing it in vacuum to form oxygen vacancies. High electron densities are obtained, at mobilities competitive with, in some cases even higher than, other methods. This offers a number of potential advantages over other doping approaches, which require the deliberate introduction of impurities. The work could have impact in oxide electronics in general, as well as in transparent conductors for a variety of devices.
Principal Investigators:
Dan Frisbie
Chris Leighton
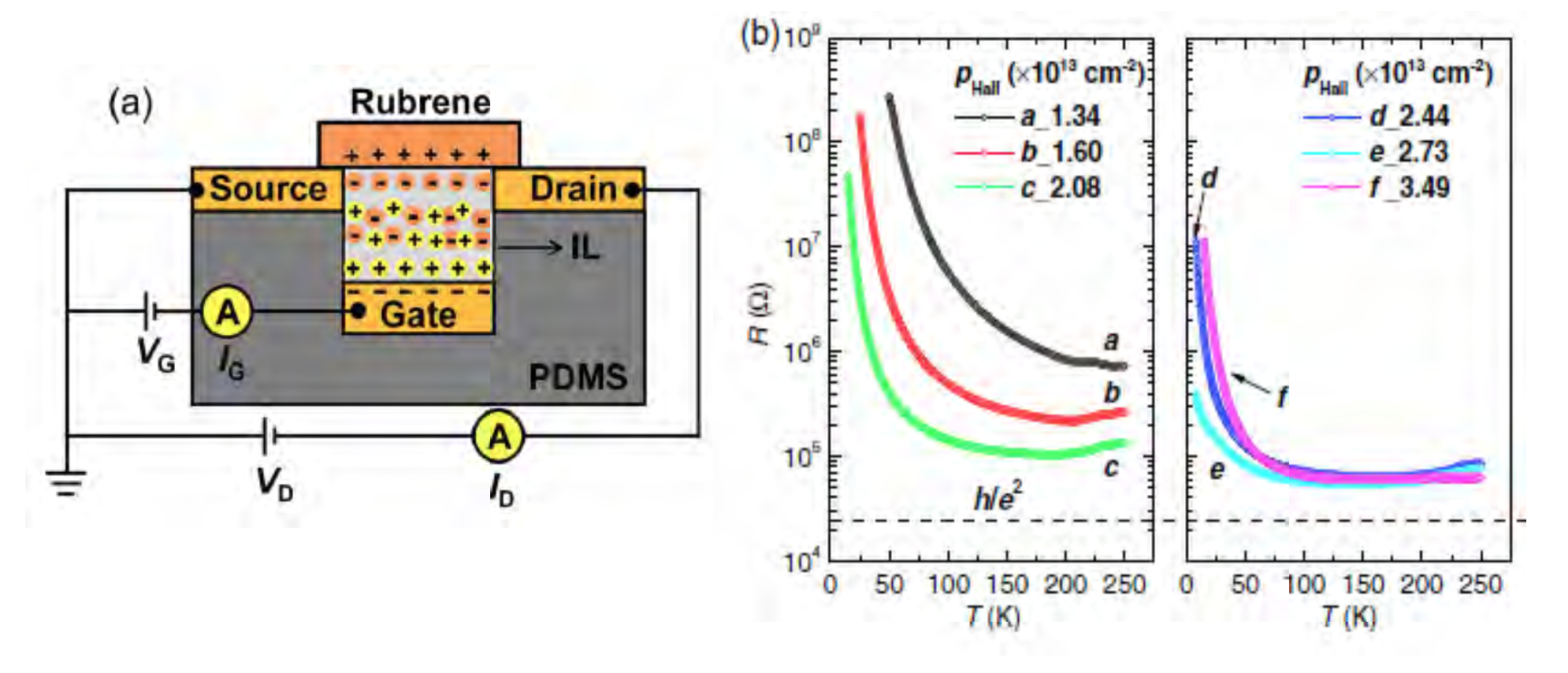
Whether metallic behavior can exist in 2D materials is a question that has troubled condensed matter physics for decades. Although originally thought impossible, evidence for such in ultra-clean high-purity doped inorganic semiconductor heterostructures based on materials such as Si and GaAs eventually changed the prevailing view. Research performed in IRG-1 using an approach to doping known as electrolyte gating has now shown that highly conductive (close to metallic) behavior can also be seen in 2D in an organic semiconductor, rubrene. This was enabled by techniques, based on the use of ionic liquids, that increase the density of holes on the surface by a thousand times over prior work. The mobility of the holes in rubrene remains far lower than inorganic semiconductors, however, raising perplexing questions about the fundamental origin of the conductive state.
Principal Investigators:
Tim Lodge
Theresa Reineke
About the figure: Change in polycationic micelle size with pH in different polyprotic buffers; hydrodyamic radius Rh measured by dynamic light scattering. Reference: J.E. Laaser, Y. Jiang, D. Sprouse, T.M. Reineke, T.P. Lodge, Macromolecules, 2015, 48, 2677-2685.
Charged nanoparticles, such as polyelectrolyte micelles, are of increasing interest in diverse applications, including gene therapy. The dimensions of these objects are critical determinants of their performance, yet their size is affected by the surroundings. In particular, we have shown that it is not just the pH that matters, but also whether the pH is established by a monoprotic or polyprotic buffer. This can be explained by a selective partitioning of polyanions (e.g., phosphate, sulfate) into the outer region of the micelle. This effect has not been documented before, but is of direct relevance to physiological conditions, where polyanions are abundant.
Education and Outreach Highlight

On May 20, 2015, over 250 middle and high school students participated in the inaugural MRSEC Research Experiences for Teachers Student Expo. The Expo extends the impact of the MRSEC RET program beyond participating teachers to their students via direct interaction with UMN researchers. During the school year, a secure website was set up to allow students to ask questions of the same researchers who mentored their teachers during the summer at UMN. After successful completion of the classroom research experience, the students were invited to the UMN campus to present their work in person via the Student Expo Poster Session. A full day of activities was planned leading up to the poster session, which included an admissions presentation, scientific demonstration show, and tours of the Minnesota Nano Center, Valspar Materials Lab, and seven faculty laboratories.
For earlier highlights, visit the NSF page, mrsec.org
UMN MRSEC
435 Amundson Hall, 421 Washington Ave. SE, Minneapolis, MN, 55455
P: 612-626-0713 | F: 612-626-7805